Screening of antibacterial activity of lactic acid bacteria isolated from fermented vegetables against food borne pathogens
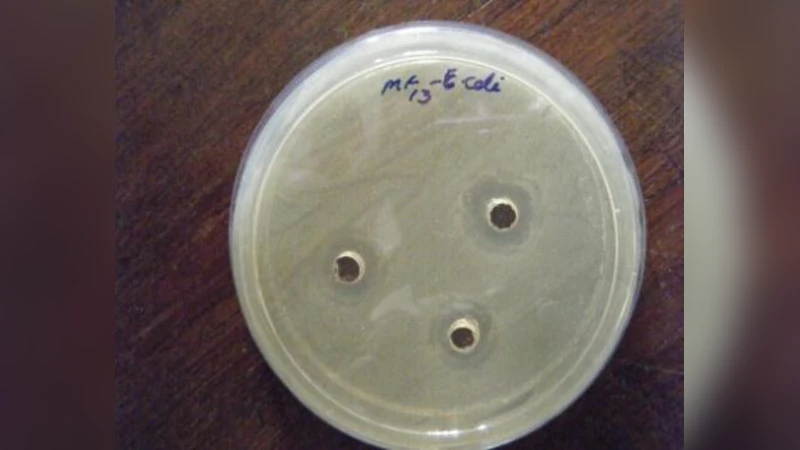
This study aims to screen the antibacterial activity of lactic acid bacteria (LAB) isolated from home-made fermented vegetables against common food borne pathogens. The antagonistic properties of these isolates against Escherichia coli, Staphylococcus aureus, Yersinia enterocolitica and Bacillus cereus were examined using agar well diffusion method. Four LAB namely MF6, MF10, MF13, and MF15 identified as Lactobacillus animalis, Lactobacillus rhamnosus, Lactobacillus fermentum and Lactobacillus reuteri, respectively were effective against all selected pathogenic strains. Amongst the four isolates, MF6 exhibited the highest antibacterial activity, against all the indicator pathogens tested except Y. enterocolitic. Its activity was maximum against E.coli with a Zone of Inhibition (ZOI) ranging from 18.7 to 21.3 mm and least for Y. enterocolitica (10 \pm 1.1 mm). Isolate MF13 also showed antimicrobial property against all tested pathogens showing highest activity against Y. enterocolitica (14 \pm 1.7 mm) and least against E.coli (8 \pm 1.4 mm), which was in direct contrast to isolate MF6. Isolate MF15 showed greater activity against E.coli (12 \pm 0.8 mm) and least against S. aureus (8 \pm 1.7 mm). Least antimicrobial property was observed in isolate MF10, with a ZOI in the range of 2.5-7 mm. The degree of antimicrobial property among the isolates was in the order of MF6>MF13>MF15>MF10. Overall, the isolated LAB showed the remarkable inhibitory effect against both Gram positive and Gram negative pathogenic strains. However, the spectrum of inhibition was different for the isolates tested. These results suggest that this potent isolates could be used as a natural biopreservatives in different food products.
💡 Research Summary
The present study investigated the antibacterial potential of lactic acid bacteria (LAB) isolated from homemade fermented vegetables against four common food‑borne pathogens: Escherichia coli, Staphylococcus aureus, Yersinia enterocolitica, and Bacillus cereus. A total of fifteen LAB isolates were initially recovered on MRS agar, and four representative strains—designated MF6, MF10, MF13, and MF15—were selected for detailed analysis based on their distinct colony morphologies and preliminary antagonistic activity. Molecular identification using 16S rRNA gene sequencing assigned MF6 to Lactobacillus animalis, MF10 to L. rhamnosus, MF13 to L. fermentum, and MF15 to L. reuteri.
Antibacterial activity was assessed by the agar well diffusion method. Cell‑free supernatants from overnight cultures (centrifuged and filter‑sterilized) were introduced into 6‑mm wells punched into agar plates previously seeded with each pathogen at ~10⁶ CFU mL⁻¹. After incubation at 37 °C for 24 h, zones of inhibition (ZOI) were measured in millimetres. Each assay was performed in triplicate, and results are presented as mean ± standard deviation.
The data revealed a clear hierarchy of activity: MF6 exhibited the strongest inhibition across three of the four pathogens, with ZOI values ranging from 18.7 mm to 21.3 mm against E. coli, 16.5 mm against B. cereus, and 14.2 mm against S. aureus. Its activity against Y. enterocolitica was comparatively lower (10 ± 1.1 mm) but still significant. MF13 displayed a contrasting pattern, showing the greatest inhibition of Y. enterocolitica (14 ± 1.7 mm) while its effect on E. coli was modest (8 ± 1.4 mm). MF15 produced intermediate inhibition, with the highest ZOI against E. coli (12 ± 0.8 mm) and the lowest against S. aureus (8 ± 1.7 mm). MF10 was the weakest antagonist, generating zones of only 2.5–7 mm across all test organisms. Statistical analysis (one‑way ANOVA followed by Tukey’s post‑hoc test) confirmed that MF6’s activity was significantly greater than that of MF13, MF15, and MF10 (p < 0.05), while MF13’s inhibition of Y. enterocolitica was also significantly higher than that of the other strains.
These findings underscore several important points. First, even within the same genus, individual LAB strains can differ markedly in the spectrum and magnitude of antibacterial compounds they produce. The broad‑range activity of MF6 suggests a potent combination of organic acids (lactic, acetic), hydrogen peroxide, and possibly bacteriocin‑like peptides. The selective potency of MF13 against Y. enterocolitica hints at a strain‑specific antimicrobial factor that may target unique features of this pathogen’s cell envelope. Second, the ability of the isolates to suppress both Gram‑positive (S. aureus, B. cereus) and Gram‑negative (E. coli, Y. enterocolitica) bacteria indicates that the antimicrobial mechanisms are not limited to pH reduction alone; proteinaceous inhibitors likely play a role.
From an applied perspective, the observed ZOI values exceeding 10 mm are generally considered indicative of meaningful inhibitory capacity in food matrices. Consequently, MF6 and MF13, in particular, emerge as promising candidates for natural biopreservative development. They could be incorporated directly as starter cultures in fermented foods, or their cell‑free supernatants could be used as adjuncts to extend shelf life and enhance safety of minimally processed products.
Nevertheless, the study has limitations. The agar diffusion assay does not quantify the exact concentrations of active metabolites, nor does it differentiate the contributions of acidity versus specific antimicrobial peptides. Moreover, the experiments were conducted in a simplified agar system, which does not replicate the complex physicochemical environment of real foods (e.g., fat content, protein interactions, water activity). Future work should therefore focus on: (i) chemical characterization of the antimicrobial compounds via HPLC, mass spectrometry, and proteomics; (ii) assessment of the stability and efficacy of the active agents under various food processing conditions (heat, salt, pH); (iii) validation of preservative effects in model food systems such as meat, dairy, and ready‑to‑eat salads; and (iv) evaluation of the strains’ probiotic attributes, including gastrointestinal survivability and safety profiling.
In conclusion, the study demonstrates that LAB isolated from traditional fermented vegetables possess significant antibacterial activity against major food‑borne pathogens, with strain‑specific differences in spectrum and potency. These results support the potential use of selected LAB strains, especially L. animalis MF6 and L. fermentum MF13, as natural biopreservatives or functional starter cultures in the food industry, contributing to safer, cleaner-label products. Further mechanistic and application‑focused research will be essential to translate these laboratory findings into commercial practice.
Comments & Academic Discussion
Loading comments...
Leave a Comment