High pressures make Hg a transition metal in a thermodynamically stable solid
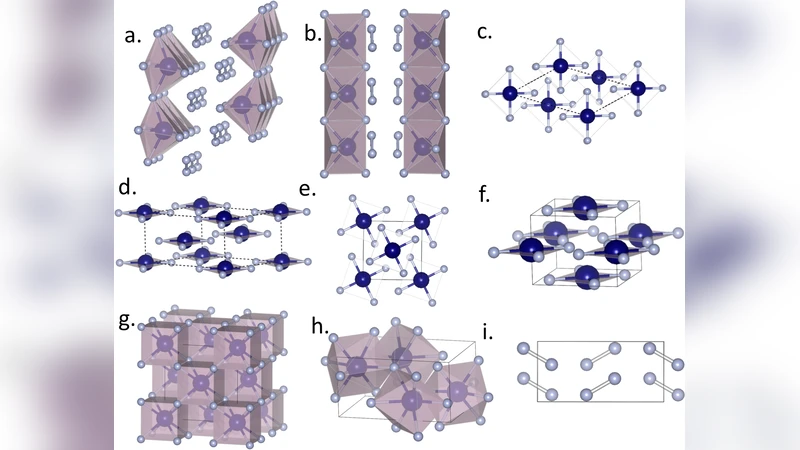
The appropriateness of including Hg among the transition metals has been debated for a long time. Although the synthesis of HgF${4}$ molecules in gas phase was reported before, the molecules show strong instabilities and dissociate. Therefore, the transition metal propensity of Hg remains an open question. Here, we propose that high pressure provides a controllable method for preparing unusual oxidation states of matter. Using an advanced structure search method based on first-principles electronic structure calculations, we predict that under high pressures, Hg can transfer the electrons in its outmost $d$ shell to F atoms, thereby acting as a transition metal. Oxidation of Hg to the +4 state yielded thermodynamically stable molecular crystals consisting of HgF${4}$ planar molecules, a typical geometry for $d^{8}$ metal centers.
💡 Research Summary
The paper tackles the long‑standing debate over whether mercury (Hg) should be classified as a transition metal. While Hg’s ground‑state electron configuration (5d¹⁰6s²) suggests a filled d‑shell and thus non‑transition‑metal behavior, gas‑phase HgF₄ molecules have been reported but are highly unstable, leaving the question unresolved. The authors propose that extreme pressure can stabilize unusual oxidation states, enabling Hg to behave as a transition metal.
Using the USPEX evolutionary algorithm, they performed a variable‑composition crystal‑structure search for the Hg–F system over a pressure range of 0–200 GPa. First‑principles density‑functional theory (DFT) calculations (PBE exchange‑correlation, PAW potentials, 600 eV cutoff) were carried out for each candidate structure. Phonon calculations verified dynamical stability, and thermodynamic stability was assessed via enthalpy and Gibbs free‑energy comparisons with competing phases (HgF₂ + ½ F₂, Hg + 2 F₂).
The results reveal that above ~50 GPa, a stoichiometric HgF₄ phase with I4/mmm (or P4/mmm) symmetry becomes the most stable compound. Its structure consists of planar square HgF₄ units: each Hg atom is four‑coordinate in a perfectly square geometry, with Hg–F bond lengths ≈1.95 Å. Projected density‑of‑states (PDOS) and charge‑density difference analyses show a substantial transfer of ≈1.8 electrons from Hg to the surrounding fluorine atoms, indicating that the 5d electrons participate in bonding. Consequently, Hg adopts a d⁸ configuration (5d⁸6s⁰) characteristic of many transition‑metal complexes, and the planar geometry is the classic coordination environment for d⁸ metal centers.
Phonon spectra contain only positive frequencies, confirming that the crystal is dynamically stable under the examined pressures. Enthalpy calculations demonstrate that HgF₄ lies below the convex hull of competing phases, making it thermodynamically favored at high pressure. The authors suggest that diamond‑anvil‑cell experiments at 50–100 GPa, combined with Raman spectroscopy (expected Hg–F modes around 300–350 cm⁻¹) and X‑ray diffraction, could verify the predicted structure.
The study illustrates a broader principle: high pressure can compress the energy gap between core‑like d orbitals and valence states, allowing elements traditionally considered non‑transition metals to access transition‑metal chemistry. By forcing Hg’s 5d electrons into covalent bonding with fluorine, pressure effectively “turns on” its transition‑metal character, stabilizing the +4 oxidation state in a solid lattice. This finding not only resolves the Hg transition‑metal controversy under extreme conditions but also opens a pathway for synthesizing other exotic oxidation states via pressure‑induced electronic reconfiguration. Future work may explore whether other post‑transition metals (e.g., Au, Ag, Pb) exhibit similar behavior, potentially expanding the periodic table’s functional landscape under high‑pressure conditions.