Can persistent Epstein-Barr virus infection induce Chronic Fatigue Syndrome as a Pavlov reflex of the immune response?
Chronic Fatigue Syndrome is a protracted illness condition (lasting even years) appearing with strong flu symptoms and systemic defiances by the immune system. Here, by means of statistical mechanics techniques, we study the most widely accepted picture for its genesis, namely a persistent acute mononucleosis infection, and we show how such infection may drive the immune system toward an out-of-equilibrium metastable state displaying chronic activation of both humoral and cellular responses (a state of full inflammation without a direct “causes-effect” reason). By exploiting a bridge with a neural scenario, we mirror killer lymphocytes $T_K$ and $B$ cells to neurons and helper lymphocytes $T_{H_1},T_{H_2}$ to synapses, hence showing that the immune system may experience the Pavlov conditional reflex phenomenon: if the exposition to a stimulus (EBV antigens) lasts for too long, strong internal correlations among $B,T_K,T_H$ may develop ultimately resulting in a persistent activation even though the stimulus itself is removed. These outcomes are corroborated by several experimental findings.
💡 Research Summary
The manuscript investigates whether a persistent Epstein‑Barr virus (EBV) infection can drive the immune system into a Pavlovian‑type conditioned state that underlies Chronic Fatigue Syndrome (CFS). The authors begin by outlining the clinical burden of CFS—characterized by debilitating fatigue lasting at least six months, cognitive dysfunction, musculoskeletal pain, and autonomic disturbances—and note its prevalence of 0.4–1 % worldwide. They highlight that a substantial proportion of CFS cases follow an acute infectious mononucleosis (AIM) episode caused by EBV, and that immunological studies consistently report elevated CD8⁺ cytotoxic T‑cell counts, abnormal cytokine profiles, and dysregulated B‑cell activity in affected patients.
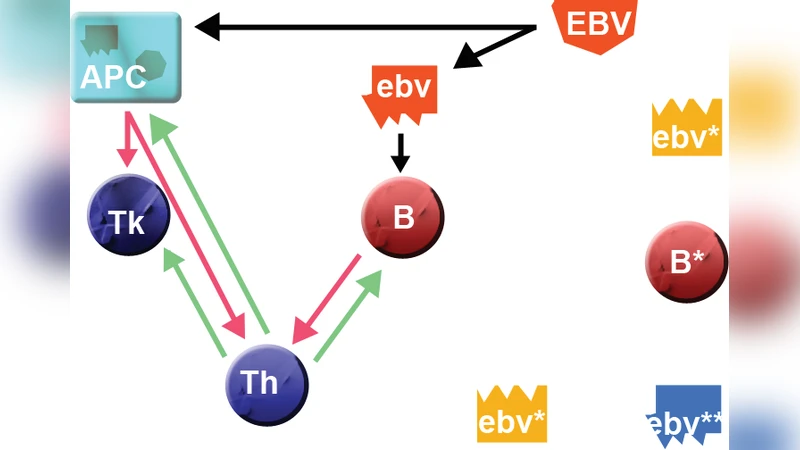
A concise immunological primer follows, describing how B cells generate antigen‑specific antibodies, how helper T cells (Th1/Th2) provide cytokine signals that activate both B cells and cytotoxic T cells (Tk), and how EBV exploits B‑cell biology. In particular, the viral lytic antigen BCRF1 mimics the human IL‑10 receptor, dampening IL‑10‑mediated immunosuppression and prolonging the acute phase. Clinical data showing that patients with a prolonged AIM phase (four months) develop a broad, high‑affinity Tk response to the early antigen BMLF1 are cited as evidence that the virus can induce a long‑lasting, self‑reinforcing immune activation.
The core of the paper is a statistical‑mechanics model that maps immune components onto a minimal neural network. Two binary “neurons” σ₁ and σ₂ represent a B‑cell clone and a Tk‑cell clone, respectively, while a single binary “synapse” J represents the population of helper T cells that modulate the interaction between them. Fast dynamics (relaxation time τ) govern the neurons, whereas the synapse evolves on a slower timescale Θ (τ ≲ Θ). External stimuli s₁ and s₂ correspond to the presence of EBV antigens that simultaneously activate B cells and Tk cells. When the stimuli are presented only for a short interval (≈τ), the synapse does not have time to strengthen; removal of either stimulus leads to immediate de‑activation of the associated neuron. Conversely, if the antigens persist for a duration comparable to Θ, Hebbian learning (w(t) = ⟨J⟩) reinforces the synapse, establishing strong correlations between σ₁ and σ₂. After this conditioning phase, removal of s₂ (or s₁) no longer silences the partner neuron because the strengthened synapse drives its activity autonomously.
Mathematically, the authors write coupled stochastic differential equations for the mean magnetizations m_B(t)=⟨σ₁⟩, m_TK(t)=⟨σ₂⟩, and the synaptic weight w(t)=⟨J⟩, derived from a master equation under the mean‑field approximation. Fixed‑point analysis reveals two metastable states: a quiescent state (low m_B, m_TK, w) and an activated state (high values of all three). The stability of the activated state depends on the ratio Θ/τ; when this ratio exceeds a critical threshold, the system inevitably settles into the activated basin, even after the external antigenic drive has vanished. Numerical simulations confirm that, under realistic parameter choices, the model reproduces the hallmark CFS immunophenotype: sustained elevation of cytotoxic Tk cells, persistent cytokine production, and a “memory” of the initial EBV exposure.
The discussion interprets these findings as a mechanistic illustration of how prolonged EBV antigen exposure can condition the adaptive immune network to maintain a chronic inflammatory response—a Pavlovian reflex of the immune system. This perspective integrates the classical conditioning paradigm with immunology, offering a unifying explanation for the paradox of ongoing immune activation in the absence of detectable viral replication. The authors suggest that therapeutic strategies aimed at disrupting the “synaptic” helper‑T‑cell coupling (e.g., cytokine blockade, modulation of T‑cell co‑stimulation pathways) could potentially de‑condition the immune network and alleviate CFS symptoms.
In summary, the paper provides a novel interdisciplinary framework that combines statistical physics, neural learning theory, and immunology to explain the transition from acute EBV infection to chronic, self‑sustaining immune activation characteristic of CFS. It bridges a gap between epidemiological observations and molecular mechanisms, and it opens new avenues for both theoretical investigation and clinical intervention.
Comments & Academic Discussion
Loading comments...
Leave a Comment