A Mathematical model for Astrocytes mediated LTP at Single Hippocampal Synapses
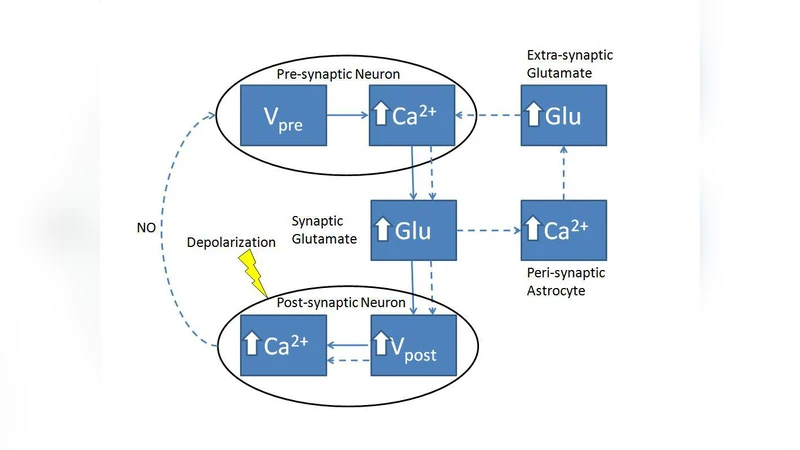
Many contemporary studies have shown that astrocytes play a significant role in modulating both short and long form of synaptic plasticity. There are very few experimental models which elucidate the role of astrocyte over Long-term Potentiation (LTP). Recently, Perea & Araque (2007) demonstrated a role of astrocytes in induction of LTP at single hippocampal synapses. They suggested a purely pre-synaptic basis for induction of this N-methyl-D- Aspartate (NMDA) Receptor-independent LTP. Also, the mechanisms underlying this pre-synaptic induction were not investigated. Here, in this article, we propose a mathematical model for astrocyte modulated LTP which successfully emulates the experimental findings of Perea & Araque (2007). Our study suggests the role of retrograde messengers, possibly Nitric Oxide (NO), for this pre-synaptically modulated LTP.
💡 Research Summary
The paper presents a comprehensive mathematical framework to explain astrocyte‑mediated, NMDA‑receptor‑independent long‑term potentiation (LTP) observed at single hippocampal Schaffer‑collateral‑CA1 synapses. Building on the experimental observations of Perea and Araque (2007), the authors construct a multi‑scale model that integrates five interacting modules: (1) pre‑synaptic action potential generation using the classic Hodgkin‑Huxley equations; (2) calcium dynamics in the presynaptic bouton, split into a fast component driven by voltage‑gated N‑type calcium channels and a slow component governed by IP₃‑dependent release from the endoplasmic reticulum, modeled with a modified Li‑Rinzel system; (3) astrocytic calcium signaling, also described by a Li‑Rinzel scheme, where glutamate binding to astrocytic mGluRs produces IP₃ and triggers calcium oscillations; (4) calcium‑dependent exocytosis of astrocytic synaptic‑like micro‑vesicles (SLMVs) that raise extracellular glutamate concentration; (5) a retrograde messenger, nitric oxide (NO), synthesized in the postsynaptic spine during mild depolarization (‑30 mV) and calcium influx through AMPA receptors, which diffuses back to the bouton and multiplicatively enhances the release probability (Pr).
The fast calcium influx is expressed as I_Ca = ρ_Ca·g_Ca·m²·(V − V_Ca), with m following Hodgkin‑Huxley kinetics. The slow calcium pool obeys differential equations for ER calcium concentration, IP₃ production (glutamate‑dependent), IP₃ degradation, and SERCA/PMCA fluxes, using Michaelis‑Menten kinetics for pumps and leaks. Parameter values are drawn from experimental measurements (e.g., bouton volume 0.13 µm³, N‑type channel density 3.2 µm⁻²) and literature.
Simulations reproduce two key experimental findings: (i) a transient increase in Pr and EPSC amplitude when astrocytes are photoreleased with caged Ca²⁺, and (ii) a persistent increase in Pr (e‑LTP) when astrocyte stimulation is paired with postsynaptic mild depolarization, even in the presence of the NMDA antagonist D‑AP5. The model shows that astrocytic Ca²⁺‑evoked glutamate release augments presynaptic mGluR activation, leading to additional calcium influx and higher Pr; the NO signal then locks this elevated Pr, providing a mechanistic basis for the observed pre‑synaptic LTP.
Compared with the earlier Nadkarni et al. (2008) model, this work adds astrocytic IP₃ signaling, extracellular glutamate feedback, and retrograde NO modulation, thereby achieving a closer quantitative match to the data. Limitations include the use of a single‑compartment representation for diffusion, reliance on assumed NO concentration‑effect relationships, and omission of protein‑synthesis dependent late‑phase LTP. The authors suggest future extensions to multi‑synapse networks and integration with live‑imaging data. Overall, the study offers a detailed, physiologically grounded computational account of how astrocytes can drive pre‑synaptic, NMDA‑independent LTP through a cascade of calcium‑mediated events and retrograde signaling.
Comments & Academic Discussion
Loading comments...
Leave a Comment