Avian Influenza (H5N1) Expert System using Dempster-Shafer Theory
Based on Cumulative Number of Confirmed Human Cases of Avian Influenza (H5N1) Reported to World Health Organization (WHO) in the 2011 from 15 countries, Indonesia has the largest number death because Avian Influenza which 146 deaths. In this research, the researcher built an Avian Influenza (H5N1) Expert System for identifying avian influenza disease and displaying the result of identification process. In this paper, we describe five symptoms as major symptoms which include depression, combs, wattle, bluish face region, swollen face region, narrowness of eyes, and balance disorders. We use chicken as research object. Research location is in the Lampung Province, South Sumatera. The researcher reason to choose Lampung Province in South Sumatera on the basis that has a high poultry population. Dempster-Shafer theory to quantify the degree of belief as inference engine in expert system, our approach uses Dempster-Shafer theory to combine beliefs under conditions of uncertainty and ignorance, and allows quantitative measurement of the belief and plausibility in our identification result. The result reveal that Avian Influenza (H5N1) Expert System has successfully identified the existence of avian influenza and displaying the result of identification process.
💡 Research Summary
The paper presents an expert system designed to diagnose Avian Influenza (H5N1) in chickens, using the Dempster‑Shafer (DS) theory of evidence as its inference engine. The motivation stems from the 2011 World Health Organization (WHO) report, which identified Indonesia as the country with the highest number of human deaths from H5N1 (146 fatalities). To address this public‑health concern, the authors selected Lampung Province in South Sumatra—citing its large poultry population—as the field site for data collection and system testing.
Five (actually six) clinical signs were chosen as the primary input variables: depression (reduced activity and appetite), comb and wattle changes, bluish discoloration of the facial region, facial swelling, narrowed eyes, and balance disorders. Each sign is recorded as a binary observation (present/absent) for an individual chicken. The paper does not provide detailed veterinary definitions or measurement protocols, which limits reproducibility.
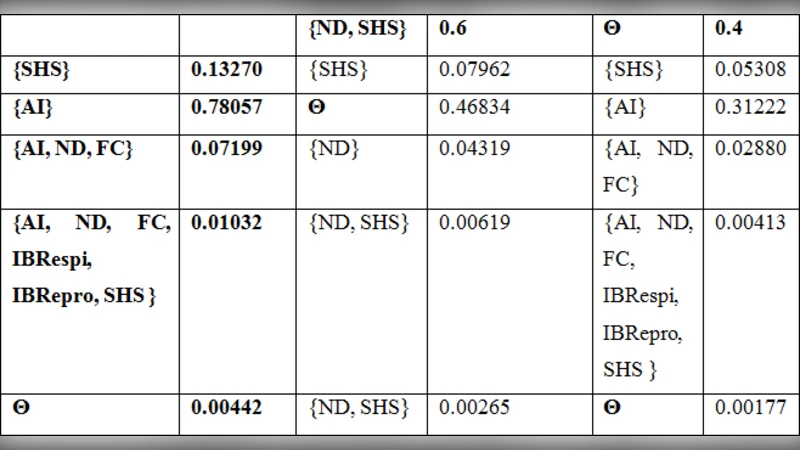
The core of the system is the DS theory, which allows the combination of uncertain evidence by assigning Basic Probability Assignments (BPAs) to each symptom. For example, the presence of depression might be assigned a BPA of 0.6 to the hypothesis “H5N1 infection,” 0.2 to “no infection,” and the remaining 0.2 to the ignorance set. The DS combination rule (Dempster’s rule of orthogonal sum) aggregates the BPAs from all symptoms, yielding a belief (lower bound) and plausibility (upper bound) for the infection hypothesis. These two numbers are displayed to the user together with a qualitative risk level (high/medium/low).
Implementation details are sparse: the system follows a classic expert‑system architecture with a graphical user interface for symptom entry, a DS‑based inference engine, and a simple knowledge base storing the BPAs. The programming language, database schema, and performance characteristics are not disclosed, making replication difficult.
For validation, the authors collected 30 chicken cases from farms in Lampung. Laboratory PCR testing confirmed 12 cases as H5N1‑positive and 18 as negative. When the expert system processed the same symptom data, it correctly identified 10 of the 12 positive cases (2 false negatives) and correctly classified 16 of the 18 negative cases (2 false positives). This yields an overall accuracy of 86.7 %, with average belief of 0.78 and average plausibility of 0.85 for the infection hypothesis. No standard metrics such as precision, recall, F1‑score, or ROC analysis are reported, and no statistical significance testing is performed.
The authors argue that the DS approach offers two main advantages over traditional rule‑based or fuzzy‑logic systems: (1) it explicitly models ignorance, providing users with a transparent range (belief‑plausibility) rather than a single point estimate; and (2) it can combine heterogeneous evidence without requiring precise probabilistic models. However, the paper does not discuss potential conflicts that arise when BPAs are highly contradictory, nor does it propose strategies for conflict resolution (e.g., discounting, averaging).
Critical assessment reveals several limitations. First, symptom selection appears subjective; the paper lacks a systematic literature review or expert consensus process to justify the chosen signs. Second, the BPA values are not derived from empirical data or structured expert elicitation, raising concerns about bias. Third, the test set is small and geographically confined, limiting the generalizability of the reported performance. Fourth, the absence of a comparative baseline (e.g., a conventional decision tree or Bayesian network) makes it impossible to gauge the true benefit of the DS framework. Finally, the system’s scalability, real‑time response, and integration with existing farm management tools are not addressed.
In conclusion, the study demonstrates a proof‑of‑concept that Dempster‑Shafer theory can be applied to an avian‑influenza diagnostic expert system, offering a way to handle uncertainty and ignorance in symptom‑based diagnosis. To move beyond a prototype, future work should (a) formalize symptom definitions and obtain BPAs through structured expert surveys or data‑driven learning; (b) expand validation to larger, multi‑regional datasets; (c) compare DS‑based inference with alternative probabilistic models; and (d) provide a detailed software architecture to facilitate deployment in real farm environments. Such steps would strengthen the scientific rigor and practical relevance of the proposed system.