Hypoxic Cell Waves around Necrotic Cores in Glioblastoma: A Biomathematical Model and its Therapeutic Implications
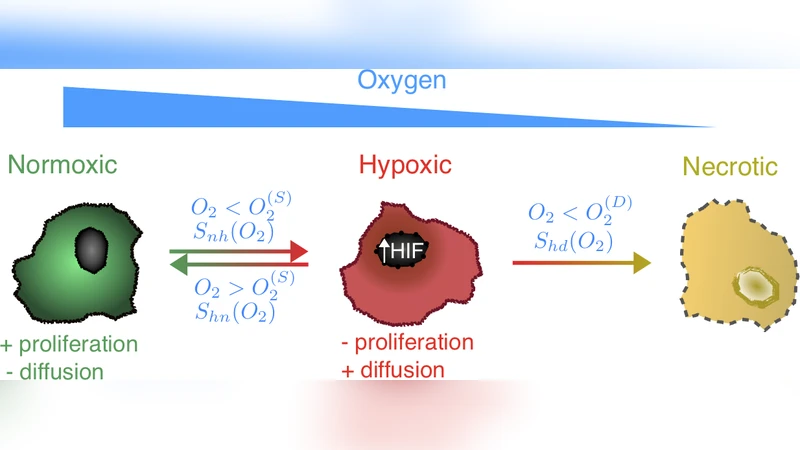
Glioblastoma is a rapidly evolving high-grade astrocytoma that is distinguished pathologically from lower grade gliomas by the presence of necrosis and microvascular hiperplasia. Necrotic areas are typically surrounded by hypercellular regions known as “pseudopalisades” originated by local tumor vessel occlusions that induce collective cellular migration events. This leads to the formation of waves of tumor cells actively migrating away from central hypoxia. We present a mathematical model that incorporates the interplay among two tumor cell phenotypes, a necrotic core and the oxygen distribution. Our simulations reveal the formation of a traveling wave of tumor cells that reproduces the observed histologic patterns of pseudopalisades. Additional simulations of the model equations show that preventing the collapse of tumor microvessels leads to slower glioma invasion, a fact that might be exploited for therapeutic purposes.
💡 Research Summary
Glioblastoma multiforme (GBM) is an aggressive high‑grade astrocytic tumor characterized by rapid proliferation, diffuse infiltration, and a distinctive histopathology that includes necrotic cores surrounded by hypercellular “pseudopalisades.” The formation of these pseudopalisades has been linked to local microvascular occlusion, which creates a hypoxic microenvironment that triggers a coordinated migratory response of tumor cells away from the necrotic zone. Despite extensive experimental documentation, a quantitative framework that integrates the underlying biological processes—cell phenotypic switching, necrosis, and oxygen transport—has been lacking. In this study, the authors develop a continuum‐based biomathematical model that captures the essential dynamics of two tumor cell phenotypes, a necrotic core, and the spatial distribution of oxygen.
The model distinguishes a proliferative phenotype that thrives under normoxic conditions and follows a logistic growth law, and a hypoxia‑induced migratory phenotype that exhibits enhanced diffusion and chemotaxis toward higher oxygen concentrations. The necrotic core is treated as a fixed region with zero oxygen supply, while surrounding healthy tissue provides oxygen through diffusion from functional microvessels. Vascular occlusion events are introduced by abruptly reducing the oxygen source term in a localized area, thereby mimicking the sudden loss of perfusion observed in vivo. The governing equations consist of coupled reaction‑diffusion–advection terms for each cell population and a diffusion‑consumption equation for oxygen. Numerical simulations are performed on a two‑dimensional lattice using finite‑difference methods, with parameter values calibrated from experimental measurements and literature reports.
Simulation results reveal that, immediately after vessel collapse, a steep oxygen gradient forms around the necrotic core. Hypoxic cells at the gradient front undergo phenotypic conversion to the migratory state, accumulate in a narrow band, and generate a traveling wave that propagates outward. The wave speed is positively correlated with the oxygen diffusion coefficient (D_O2), the cellular motility coefficient (D_c), and the intensity of vascular occlusion (α). Parameter sweeps demonstrate that eliminating vessel collapse (α ≈ 0) suppresses wave formation and markedly slows overall tumor expansion, whereas increasing α accelerates wave propagation, thickens the pseudopalisade, and amplifies tumor volume growth. These findings quantitatively reproduce the histological thickness and spatial organization of pseudopalisades observed in patient specimens, thereby validating the model’s biological relevance.
From a therapeutic perspective, the model predicts that interventions aimed at preserving microvascular integrity—such as angiogenic stabilizers, anti‑thrombotic agents, or agents that improve oxygen delivery—can diminish the hypoxia‑driven migratory wave, consequently reducing the invasive front of GBM. The authors propose that targeting the vascular collapse pathway may complement existing cytotoxic and anti‑angiogenic strategies, offering a novel avenue to delay tumor spread. They also outline future directions, including the integration of additional microenvironmental factors (e.g., extracellular matrix stiffness, immune cell infiltration) and the application of optimal control theory to design patient‑specific treatment schedules based on model predictions. In summary, this work provides a mechanistic, mathematically rigorous explanation for pseudopalisade formation and highlights microvascular protection as a promising therapeutic target in glioblastoma management.
Comments & Academic Discussion
Loading comments...
Leave a Comment