Immunity to Plasmodium knowlesi H strain malaria in olive baboons
Malaria disease is a major global health and economic development threat. It results in approximately 2.7 million deaths annually. There is currently no vaccine that has been licensed for use against malaria. Studies in animal models, especially non-human primates can lead to the revelation of possible immunological mechanisms that can lead to protection or predisposition of the host to malaria. Plasmodium knowlesi, a simian and human malaria parasite, is an attractive experimental parasite for malaria research since it can infect olive baboons (Papio anubis), non-human primates that have similar host-pathogen interactions to humans. This study was carried out to determine host immunological profiles provoked in olive baboons during the course of an infection with Plasmodium knowlesi. A total of eight adult baboons were intravenously inoculated with overnight cultured blood stage P. knowlesi H strain parasites. Five of these baboons became acutely infected while the other three became chronically infected. The immunological basis of this dual outcome of the infection was determined by measuring circulating cytokine (T helper 1 and T helper 2) and antibody (immunoglobulin G and immunoglobulin M) responses elicited in the infected baboons on a weekly basis by Enzyme Linked Immunosorbent Assay (ELISA) for up to six weeks post infection. Generated data for the first time indicated that acute P. knowlesi malaria is accompanied by increased concentrations of interferon gamma (IFN gamma), tumour necrosis factor alpha (TNF alpha) and IL 6 and reduced levels of circulating interleukin 10 (IL 10), IL 4, IL 12, immunoglobulin G (IgG) and IgM in the baboon host. These results are largely agreeable with data from human studies, thereby increasing the relevance of the olive baboon - P. knowlesi experimental infection system for future malaria studies.
💡 Research Summary
This study investigated the immunological differences between acute and chronic Plasmodium knowlesi infections in the olive baboon (Papio anubis), an established non‑human primate model that closely mimics human malaria. Eight adult baboons were intravenously inoculated with 1 × 10⁶ overnight‑cultured blood‑stage P. knowlesi H‑strain parasites. Over a six‑week period, weekly blood samples were collected and serum concentrations of key T‑helper 1 (Th1) and T‑helper 2 (Th2) cytokines—interferon‑γ (IFN‑γ), tumor necrosis factor‑α (TNF‑α), interleukin‑6 (IL‑6), interleukin‑10 (IL‑10), interleukin‑4 (IL‑4), and interleukin‑12 (IL‑12)—as well as immunoglobulin G (IgG) and immunoglobulin M (IgM) were quantified by enzyme‑linked immunosorbent assay (ELISA).
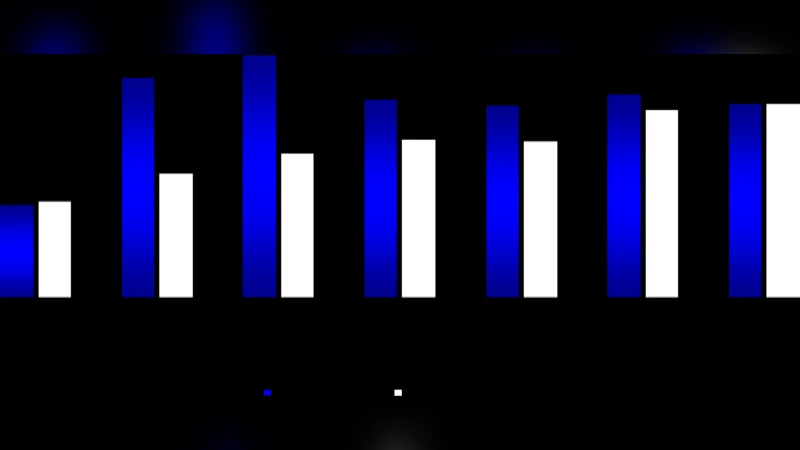
Five baboons developed an acute disease phenotype characterized by high parasitemia (>5 % of red blood cells), fever, anemia, and marked weight loss. In this group, serum IFN‑γ, TNF‑α, and IL‑6 rose sharply, peaking at approximately 250 pg/mL, 180 pg/mL, and 120 pg/mL respectively by week two. Conversely, anti‑inflammatory IL‑10 and Th2‑associated IL‑4 and IL‑12 fell to low levels (IL‑10 <5 pg/mL). IgG and IgM concentrations also declined during the acute phase, suggesting that the humoral response was either consumed or suppressed while the innate inflammatory response dominated.
The remaining three baboons exhibited a chronic, low‑parasitemia infection (<1 % parasitemia) with minimal clinical signs. Their cytokine profile was essentially the opposite of the acute group: IFN‑γ and TNF‑α remained modest (≈80 pg/mL and 60 pg/mL), IL‑6 was only mildly elevated, while IL‑10, IL‑4, and IL‑12 were sustained at relatively high levels (IL‑10 ≈30 pg/mL). Importantly, IgG and IgM levels recovered and even exceeded baseline values (IgG up to 18 µg/mL, IgM up to 12 µg/mL by week six), indicating an active, effective humoral response that likely contributed to parasite control without excessive inflammation.
These findings delineate two distinct immunological pathways governing the outcome of P. knowlesi infection in baboons. Acute infection is driven by a Th1‑biased, pro‑inflammatory cascade (high IFN‑γ, TNF‑α, IL‑6) that attempts rapid parasite clearance but can cause host tissue damage when unregulated. Chronic infection, by contrast, is characterized by a balanced immune environment where regulatory IL‑10 and Th2 cytokines limit inflammation, and robust antibody production provides long‑term control of the parasite.
The cytokine and antibody patterns observed align closely with data from human malaria studies: patients with severe or acute Plasmodium infection typically display elevated IFN‑γ, TNF‑α, and IL‑6 alongside reduced IL‑10 and antibody titers, whereas convalescent or chronically exposed individuals show restored IL‑10 and IgG/IgM levels. This concordance validates the olive baboon–P. knowlesi model as a translational platform for dissecting human malaria immunity.
From a translational perspective, the study suggests that therapeutic strategies for acute malaria should incorporate agents that temper the cytokine storm (e.g., anti‑TNF or IL‑6 blockade) while preserving parasite‑killing mechanisms. Conversely, vaccine designs aiming for durable protection may benefit from formulations that promote strong, sustained antibody responses and regulatory cytokine production, mirroring the chronic infection profile.
In summary, the research provides the first longitudinal, comparative immunoprofiling of acute versus chronic P. knowlesi infection in a non‑human primate. It demonstrates that the balance between pro‑inflammatory Th1 responses and regulatory/antibody‑mediated Th2 pathways determines disease severity. The data reinforce the relevance of the baboon model for pre‑clinical testing of malaria vaccines, immunomodulatory drugs, and novel therapeutics, and lay the groundwork for future investigations into immune memory, reinfection dynamics, and host‑parasite co‑evolution.
Comments & Academic Discussion
Loading comments...
Leave a Comment