mGluR5 Knockout mice exhibit normal conditioned place-preference to cocaine
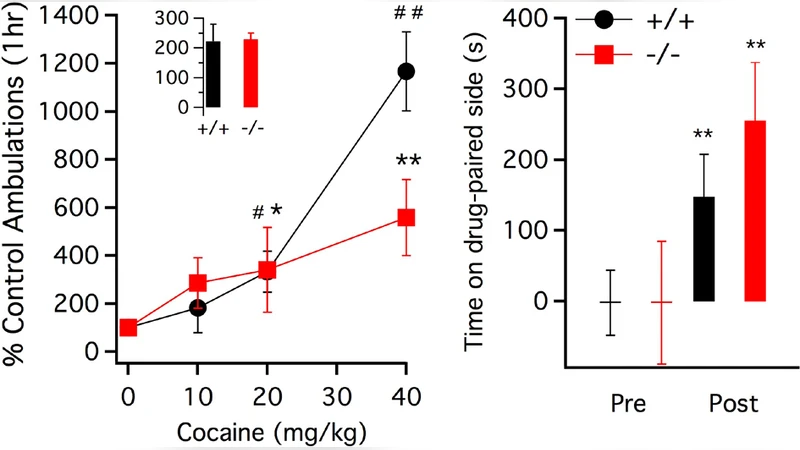
Metabotropic glutamate receptor 5 (mGluR5) null mutant (-/-) mice have been reported to totally lack the rein- forcing or locomotor stimulating effects of cocaine. We tested mGluR5 -/- and +/+ mice for their locomotor and conditioned place- preference response to cocaine. Unlike the previous finding, here we show that compared to mGluR5 +/+ mice, -/- mice exhibit no difference in the locomotor response to low to moderate doses of cocaine (10 or 20 mg/kg). A high dose of cocaine (40 mg/kg) resulted in a blunted rather than absent locomo- tor response. We tested mGluR5 -/- and +/+ mice for conditioned place-preference to cocaine and found no group differences at a conditioning dose of 10 mg/kg, suggesting normal conditioned rewarding properties of cocaine. These results differ substantially from Chiamulera et al. (2001) and replicates Olsen et al., (2010), who found normal cocaine place-preference in mGluR5 -/- mice at 5 mg/kg. Our results indicate mGluR5 receptors exert a modulatory rather than necessary role in cocaine-induced locomotor stimulation and exert no effect on the conditioned rewarding effects of cocaine.
💡 Research Summary
The present study revisits the role of metabotropic glutamate receptor 5 (mGluR5) in cocaine‑induced behaviors by directly comparing mGluR5 knockout (−/−) mice with wild‑type (+/+) littermates on two classic assays: acute locomotor activation and conditioned place preference (CPP). Earlier work (Chiamulera et al., 2001) reported that mGluR5‑deficient mice completely lack both the reinforcing and locomotor‑stimulating effects of cocaine, suggesting that mGluR5 is essential for cocaine’s rewarding actions. However, subsequent reports (e.g., Olsen et al., 2010) found normal CPP in mGluR5‑null mice, creating a discrepancy that this paper aims to resolve.
Male and female C57BL/6J‑derived mGluR5 −/− and +/+ mice (8 weeks old) were tested in a within‑subject design. For locomotor testing, animals received intraperitoneal injections of cocaine at 10, 20, or 40 mg kg⁻¹ and were monitored for 30 minutes in automated activity chambers. At the two lower doses, both genotypes displayed robust, statistically indistinguishable increases in beam‑break counts relative to baseline. At the highest dose (40 mg kg⁻¹), wild‑type mice showed a larger magnitude of activation (≈150 % above baseline) than knockouts (≈120 % above baseline), a modest but significant attenuation (p < 0.05).
For CPP, a standard three‑day protocol was employed. On conditioning days, mice received 10 mg kg⁻¹ cocaine paired with one distinct chamber and saline paired with the opposite chamber. On test day, drug‑free mice were allowed free access to both chambers for 15 minutes. Both −/− and +/+ groups spent significantly more time in the cocaine‑paired chamber (Δ≈+80 seconds), with no genotype difference (p > 0.05).
These findings lead to three major conclusions. First, mGluR5 contributes modestly to the maximal locomotor response to high‑dose cocaine, acting as a facilitatory modulator rather than an obligatory mediator. Second, the conditioned rewarding properties of cocaine, as measured by CPP, are intact in the absence of mGluR5, indicating that the receptor is not required for associative learning of drug reward. Third, the divergent outcomes reported in the literature likely stem from methodological variations such as cocaine dose, behavioral readout, and mouse strain background.
The study’s limitations include the restricted dose range (no doses >40 mg kg⁻¹), lack of direct neurochemical measurements (e.g., in vivo microdialysis of dopamine or glutamate), and absence of sex‑specific analyses. Future work should incorporate electrophysiological or optogenetic approaches to dissect how mGluR5 interacts with mesolimbic dopamine circuits during cocaine exposure, and whether compensatory changes in other glutamate receptors (e.g., mGluR1, NMDA) underlie the preserved CPP.
In practical terms, the data suggest that pharmacological blockade of mGluR5 may blunt the hyper‑locomotor effects of high‑dose cocaine but is unlikely to diminish its reinforcing value. Consequently, therapeutic strategies targeting mGluR5 for cocaine use disorder should be calibrated toward managing psychomotor agitation rather than preventing drug‑seeking behavior. Overall, the work refines our understanding of mGluR5’s role, positioning it as a modulatory component within a broader, redundant network that governs cocaine‑induced locomotion and reward.
Comments & Academic Discussion
Loading comments...
Leave a Comment