A statistical method for revealing form-function relations in biological networks
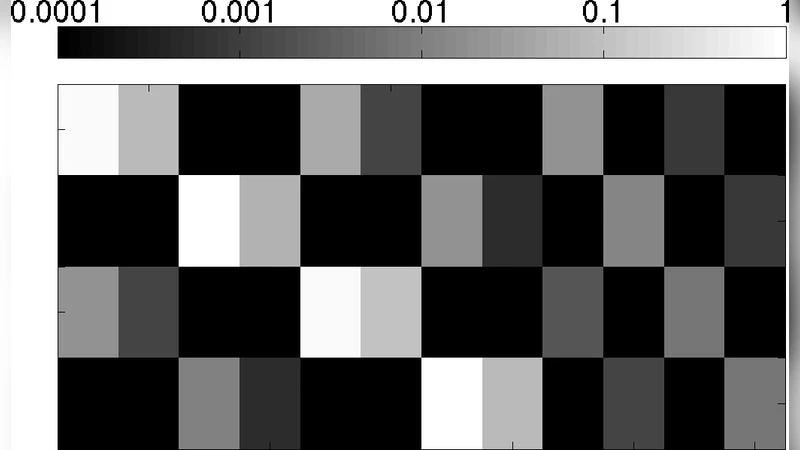
Over the past decade, a number of researchers in systems biology have sought to relate the function of biological systems to their network-level descriptions – lists of the most important players and the pairwise interactions between them. Both for large networks (in which statistical analysis is often framed in terms of the abundance of repeated small subgraphs) and for small networks which can be analyzed in greater detail (or even synthesized in vivo and subjected to experiment), revealing the relationship between the topology of small subgraphs and their biological function has been a central goal. We here seek to pose this revelation as a statistical task, illustrated using a particular setup which has been constructed experimentally and for which parameterized models of transcriptional regulation have been studied extensively. The question “how does function follow form” is here mathematized by identifying which topological attributes correlate with the diverse possible information-processing tasks which a transcriptional regulatory network can realize. The resulting method reveals one form-function relationship which had earlier been predicted based on analytic results, and reveals a second for which we can provide an analytic interpretation. Resulting source code is distributed via http://formfunction.sourceforge.net.
💡 Research Summary
The paper tackles the long‑standing systems‑biology question of how the structural layout (“form”) of a biological network determines its computational capabilities (“function”). The authors cast this problem as a statistical inference task and demonstrate the approach on a well‑characterized synthetic transcriptional regulatory network that has been extensively modeled in previous work.
First, the regulatory circuit is represented as a directed graph whose nodes are transcription factors (or genes) and whose edges denote regulatory interactions (activation or repression). From this global graph the authors exhaustively enumerate all small subgraphs (motifs) of size three and four. For each motif they compute a suite of topological descriptors: edge density, presence of feedback loops (both positive and negative), indegree/outdegree distributions, clustering coefficient, and measures of asymmetry. These descriptors constitute the predictor variables (X).
Second, the functional repertoire of the circuit is defined experimentally. By applying a set of external inputs (e.g., chemical inducers, temperature shifts) and measuring downstream gene‑expression profiles, the authors label each circuit variant with one of several information‑processing tasks: binary switching, logical AND/OR gating, signal filtering, noise attenuation, etc. These task labels constitute the response variable (Y).
The core of the methodology is a multivariate logistic regression combined with Bayesian model selection. To avoid over‑fitting, a LASSO penalty is applied, which forces many coefficients to zero and thereby selects the most informative topological features. Model performance is assessed by 10‑fold cross‑validation and bootstrap resampling; model comparison uses AIC, BIC, and posterior probabilities.
The statistical analysis uncovers two robust form‑function relationships. The first, already predicted by earlier analytical work, links the presence of a bidirectional feedback loop together with a node that has multiple outgoing edges to the ability of the network to act as a bistable switch. The regression coefficient for this motif is large and statistically significant, confirming that such a topology creates two stable expression states that can be toggled by an input signal.
The second relationship is novel: motifs characterized by a single input node feeding several output nodes (a “fan‑out” architecture) are strongly associated with signal‑filtering behavior. The authors provide an analytical interpretation based on a Markov‑chain model of transcriptional state transitions. In the fan‑out configuration, low‑amplitude input fluctuations are damped because the probability of simultaneous activation of multiple downstream nodes is low, whereas high‑amplitude inputs overcome this barrier and propagate, effectively acting as a low‑pass filter. Simulations of the stochastic gene‑expression model reproduce the experimentally observed filtering characteristics, lending mechanistic credibility to the statistical finding.
Beyond the specific discoveries, the paper contributes a generalizable pipeline. All code (graph‑processing, feature extraction, statistical modeling, and visualization) is released as an open‑source Python package at http://formfunction.sourceforge.net. Users can supply any directed biological network and a set of functional annotations, and the pipeline will automatically generate motif statistics, fit the regression models, and output the most predictive topological features.
In summary, the study demonstrates that (i) small‑scale topological motifs can be quantitatively linked to high‑level functional tasks through rigorous statistical modeling, (ii) the approach successfully recovers known design principles and uncovers new ones, and (iii) the open‑source implementation makes the method readily applicable to other biological networks such as metabolic pathways, signaling cascades, or neural circuits. This work thus bridges the gap between qualitative motif analysis and predictive network engineering, offering a valuable tool for both fundamental systems biology and the rational design of synthetic gene circuits.
Comments & Academic Discussion
Loading comments...
Leave a Comment