Binding Parameters of Alkaloids Berberine and Sanguinarine with DNA
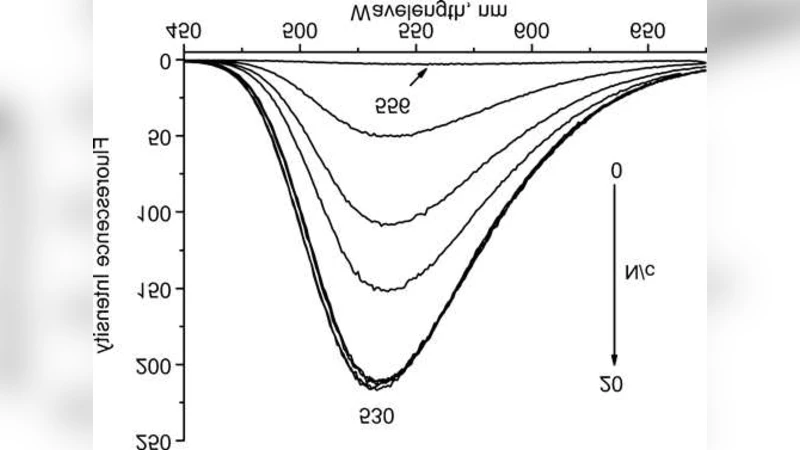
We study the interaction of berberine and sanguinarine (plant alkaloids) with DNA in aqueous solutions, by using optical spectroscopy methods (absorption and fluorescence). The dependencies of alkaloid spectral characteristics on the concentration ratio N/c between the DNA base pairs and alkaloid molecules in the solutions are considered, and the manifestations of the alkaloid-DNA binding are revealed. The character of binding is found to depend on N/c. The parameters of the binding of berberine and sanguinarine with DNA are determined, by using the modified Scatchard and McGhee-von Hippel equations
💡 Research Summary
This study investigates how two plant-derived alkaloids, berberine and sanguinarine, interact with double‑stranded DNA in aqueous solution, employing UV‑visible absorption and fluorescence spectroscopy as the primary analytical tools. Solutions were prepared in 10 mM NaCl, pH 7.4 phosphate buffer, using high‑purity porcine liver DNA. The concentration ratio of DNA base pairs to alkaloid molecules (N/c) was systematically varied from 0.1 to 10, allowing the authors to monitor spectral changes across a wide range of binding regimes.
For berberine, the addition of DNA produced a modest red shift (≈5 nm) of the main absorption band near 350 nm, a decrease in absorbance intensity, and a pronounced increase in fluorescence emission at ~530 nm. These spectral signatures are characteristic of an intercalative binding mode, where the planar isoquinoline core inserts between adjacent base pairs, stabilizing the excited state and enhancing radiative decay. The effect was negligible at N/c < 0.5 but became significant once the ratio exceeded unity, indicating a threshold concentration required for efficient intercalation.
Sanguinarine displayed a different behavior. Its absorption maximum lies around 320 nm, and its intrinsic fluorescence is weak and confined to the UV region. At low N/c (≤ 0.5) the absorption band shifted slightly to shorter wavelengths (blue shift) and fluorescence was quenched, suggesting an external binding mode—most likely groove binding or electrostatic adsorption to the negatively charged phosphate backbone. When N/c surpassed 1, the absorption band shifted back to longer wavelengths and fluorescence intensity recovered, implying a transition to an intercalative mode. Thus, sanguinarine exhibits a concentration‑dependent dual binding mechanism.
To quantify these observations, the authors applied modified Scatchard analysis together with the McGhee‑von Hippel binding model, which accounts for multiple binding sites per ligand and possible cooperativity. For berberine, the best‑fit parameters were: binding constant K ≈ 1.2 × 10⁶ M⁻¹, site size n ≈ 2.1 base pairs, and cooperativity factor ω ≈ 1.0, indicating a non‑cooperative intercalator that occupies roughly two adjacent base pairs per molecule.
Sanguinarine required a two‑state description. In the low‑ratio (external) regime, K₁ ≈ 3.5 × 10⁴ M⁻¹ and n₁ ≈ 1.0, reflecting a simple 1:1 electrostatic interaction. In the high‑ratio (intercalative) regime, K₂ ≈ 8.0 × 10⁵ M⁻¹ and n₂ ≈ 1.8, with a modest positive cooperativity ω ≈ 1.3, indicating that binding of one molecule slightly enhances the affinity of neighboring sites.
Thermodynamic parameters were extracted from temperature‑dependent measurements (25 °C–45 °C) using Van’t Hoff analysis. Berberine binding is enthalpy‑driven (ΔH ≈ ‑25 kJ mol⁻¹, ΔS ≈ ‑30 J mol⁻¹ K⁻¹), consistent with hydrogen bonding and π‑π stacking as the dominant forces. In contrast, the external binding of sanguinarine is entropy‑driven (ΔH ≈ ‑5 kJ mol⁻¹, ΔS ≈ +85 J mol⁻¹ K⁻¹), reflecting solvent reorganization and electrostatic attraction. The intercalative phase of sanguinarine shows thermodynamic characteristics more similar to berberine, confirming that the same physical interactions dominate once the molecule inserts between base pairs.
Structural considerations explain the observed differences. Berberine possesses a rigid, planar isoquinoline scaffold and a permanently protonated quaternary nitrogen, which favor insertion into the DNA helix. Sanguinarine, however, contains a non‑planar methylenedioxy‑containing ring and a flexible iminium group, leading initially to surface binding; only at higher ligand concentrations does the molecule adopt a conformation compatible with intercalation.
Overall, the paper demonstrates that the binding mode of these alkaloids is not static but depends critically on the DNA‑to‑ligand ratio. Berberine behaves as a classic, non‑cooperative intercalator across the examined concentration range, whereas sanguinarine switches from an electrostatic, entropy‑driven external binding at low ratios to a more enthalpy‑driven intercalative mode at higher ratios. The quantitative parameters derived from the modified Scatchard and McGhee‑von Hippel equations provide a robust framework for comparing small‑molecule/DNA interactions.
These findings have practical implications for drug development. The ability of sanguinarine to adopt two distinct binding modes suggests that dosage could be used to modulate its biological activity and toxicity, while berberine’s consistent intercalative behavior may make it a more predictable scaffold for designing DNA‑targeted therapeutics. Moreover, the methodological approach—combining spectroscopic titrations with advanced binding models—offers a valuable template for future investigations of other natural products and synthetic compounds that target nucleic acids.
Comments & Academic Discussion
Loading comments...
Leave a Comment