Maladaptation and the paradox of robustness in evolution
Background. Organisms use a variety of mechanisms to protect themselves against perturbations. For example, repair mechanisms fix damage, feedback loops keep homeostatic systems at their setpoints, and biochemical filters distinguish signal from noise. Such buffering mechanisms are often discussed in terms of robustness, which may be measured by reduced sensitivity of performance to perturbations. Methodology/Principal Findings. I use a mathematical model to analyze the evolutionary dynamics of robustness in order to understand aspects of organismal design by natural selection. I focus on two characters: one character performs an adaptive task; the other character buffers the performance of the first character against perturbations. Increased perturbations favor enhanced buffering and robustness, which in turn decreases sensitivity and reduces the intensity of natural selection on the adaptive character. Reduced selective pressure on the adaptive character often leads to a less costly, lower performance trait. Conclusions/Significance. The paradox of robustness arises from evolutionary dynamics: enhanced robustness causes an evolutionary reduction in the adaptive performance of the target character, leading to a degree of maladaptation compared to what could be achieved by natural selection in the absence of robustness mechanisms. Over evolutionary time, buffering traits may become layered on top of each other, while the underlying adaptive traits become replaced by cheaper, lower performance components. The paradox of robustness has widespread implications for understanding organismal design.
💡 Research Summary
Steven A. Frank’s 2007 PLoS ONE paper tackles a counter‑intuitive phenomenon he calls the “paradox of robustness.” The central idea is that mechanisms which buffer organisms against environmental or developmental perturbations—what biologists refer to as robustness or safety factors—can, over evolutionary time, reduce the selective pressure on the traits they protect. When selection weakens, those protected traits tend to evolve toward cheaper, lower‑performance versions because the cost of maintaining high performance is no longer justified. Consequently, the organism becomes maladapted relative to the optimum it could achieve in the absence of any buffering.
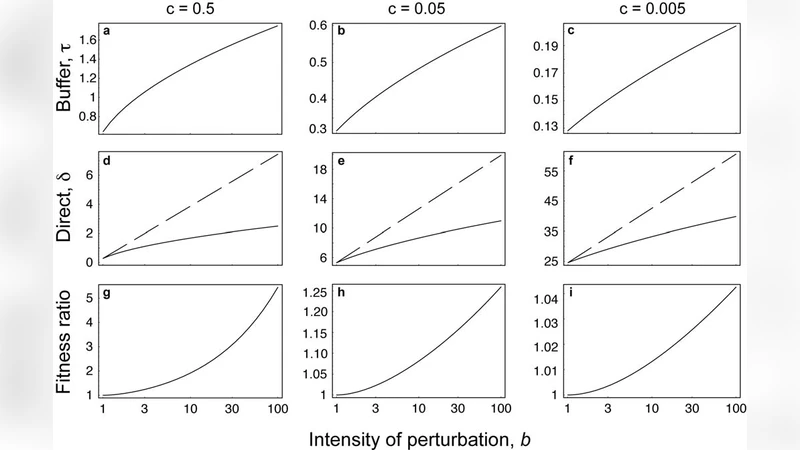
To formalize this intuition, Frank builds a simple two‑character model. One character (d) performs an adaptive function (e.g., discriminating correct from incorrect ligands), while a second character (t) acts as a buffer that reduces the sensitivity of the first character to perturbations. The fitness function incorporates benefits from correct signaling, penalties from false signals, and explicit costs for both characters (c·d for the adaptive trait and b·t for the buffer). The parameter b captures the intensity of external perturbations (the cost of false signals), and c reflects the intrinsic cost of improving discrimination.
Using kinetic proofreading as a concrete example, Frank shows that increasing the buffer (t) lowers the probability that any ligand triggers a signal, thereby reducing false positives more than true positives. However, this same reduction in signal probability also diminishes the marginal benefit of improving discrimination (d). By numerically solving for the joint optimum (d*, t*) under varying b and c, he demonstrates a clear pattern: as perturbation intensity rises, the optimal buffer grows while the optimal adaptive trait shrinks. When the buffer is held fixed at a low value, the adaptive trait would evolve to a much higher performance level, but the co‑evolution of the buffer prevents this, leading to a measurable loss of performance—quantified by the logarithmic ratio of false‑signal probabilities between the constrained and unconstrained scenarios.
Frank further links this dynamic to genetic robustness. A stronger buffer reduces the effective selection coefficient on d, allowing deleterious mutations in d to persist longer. This slows the purging of harmful alleles and can increase standing genetic variation, echoing classic arguments about mutation‑selection balance. Yet, unlike purely genetic robustness discussions, Frank emphasizes the cost‑benefit trade‑off: buffers are themselves costly, and their evolution is driven by the need to offset perturbation costs, not merely by mutation pressure.
The paper culminates in a broader evolutionary narrative. Robustness mechanisms tend to accumulate in layers—new buffers are added on top of older ones—while the underlying adaptive traits are progressively replaced by cheaper, less precise versions. This “layered buffering” explains why many biological systems exhibit redundant safety factors (e.g., multiple DNA repair pathways, overlapping metabolic controls) and why core functional components often appear sub‑optimal when examined in isolation.
Implications are wide‑ranging. In physiology, the presence of excess lung capacity or bone strength may reflect historical buffering that allowed the underlying tissue to become less robust. In molecular biology, kinetic proofreading, error‑correcting polymerases, and chaperone networks can be seen as buffers that permit the primary catalytic domains to evolve with reduced fidelity. In evolutionary theory, the paradox of robustness challenges the assumption that robustness always enhances organismal performance; instead, it can drive a hidden form of maladaptation.
Frank’s analysis thus bridges two research traditions—cost‑benefit safety‑factor models and genetic robustness theory—by focusing on the evolutionary dynamics of two interacting traits. The resulting framework provides a quantitative tool for measuring robustness (via sensitivity reduction) and maladaptation (via performance loss relative to a no‑buffer optimum). It invites researchers to re‑examine the design of biological systems, recognizing that the very mechanisms that protect life may also be the agents of its subtle, long‑term inefficiencies.
Comments & Academic Discussion
Loading comments...
Leave a Comment