The effect of point mutations on energy conduction pathways in proteins
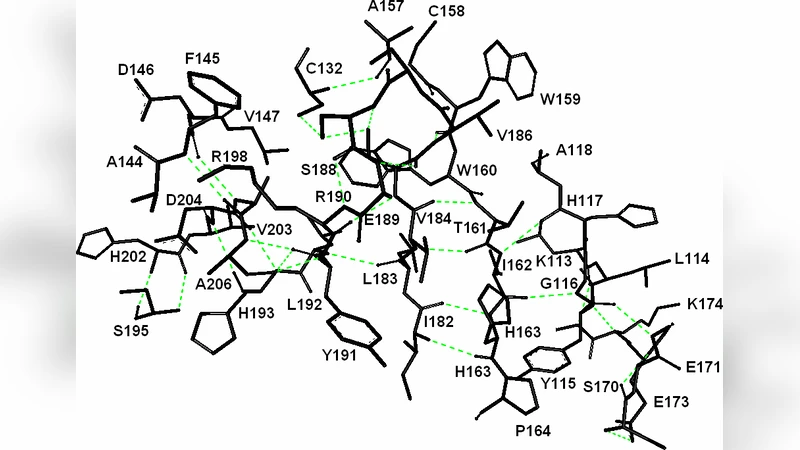
Energetically responsive residues of the 173 amino acid N-terminal domain of the cardiac Ryanodine receptor RyR2 are identified by a simple elastic net model. Residues that respond in a correlated way to fluctuations of spatially neighboring residues specify a hydrogen bonded path through the protein. The evolutionarily conserved residues of the protein are all located on this path or in its close proximity. All of the residues of the path are either located on the two Mir domains of the protein or are hydrogen bonded them. Two calcium binding residues, E171 and E173, are proposed as potential binding region, based on insights gained from the elastic net analysis of another calcium channel receptor, the inositol 1,4,5-triphosphate receptor, IP3R. Analysis of the disease causing A77V mutated RyR2 showed that the path is disrupted by the loss of energy responsiveness of certain residues.
💡 Research Summary
The paper investigates how point mutations affect energy conduction pathways in the cardiac ryanodine receptor RyR2, focusing on the N‑terminal domain (173 residues). Using a simple elastic net model—a coarse‑grained, harmonic network approach—the authors quantify the energetic response of each residue (U_i) to thermal fluctuations and compute pairwise energy correlations (U_ij). Residues with high U_i values and strong U_ij links form a continuous “energy conduction pathway” that transmits conformational information across the protein.
Applying this method to the wild‑type RyR2 structure (PDB 3IM5), the authors find that the pathway residues largely overlap with evolutionarily conserved residues identified in previous studies. All pathway residues are either located within the two Mir domains (residues 124‑178 and 164‑217) or are hydrogen‑bonded to them, suggesting that the Mir domains serve as a structural scaffold for energy transport. Notably, two glutamic acids, E171 and E173, sit at the distal end of the pathway and display strong energetic responsiveness; by analogy with the inositol 1,4,5‑triphosphate receptor (IP3R), where E283 and E285 form a calcium‑binding site, the authors propose that E171/E173 may constitute a calcium‑binding region in RyR2.
The disease‑causing A77V mutation replaces a small alanine with a bulkier valine, introducing two extra methyl groups that perturb the local packing. By comparing inter‑residue distance matrices (ΔR_ij) between wild‑type (3IM5) and mutant (3IM7) structures, the study shows that a large fraction of the 15,051 residue‑pair distances change beyond the experimental resolution (2.2–2.5 Å). These perturbations are not confined to the immediate vicinity of residue 77; instead, they propagate throughout the protein and specifically disrupt the energetic coupling of several key pathway residues (e.g., residues 45, 78, 112, etc.). In the mutant, these residues lose their high U_i values and strong U_ij correlations, effectively breaking the energy conduction pathway, as illustrated by the red‑highlighted residues in Figure 6.
To validate the generality of their findings, the authors repeat the elastic net analysis on the IP3R structure (PDB 1N4K). The resulting energy‑responsive residues again map onto the Mir‑like domains and overlap with conserved residues, reinforcing the notion that Mir domains are universal hubs for energy transmission in calcium‑release channels. Moreover, the IP3 ligand binds near residue R265, which lies on the calculated energy‑correlation path, while the calcium‑binding glutamates (E283/E285) sit at the path’s terminus, mirroring the RyR2 situation.
In discussion, the authors argue that the elastic net model, despite its simplicity, captures biologically relevant energy pathways that are evolutionarily conserved and structurally anchored by Mir domains. The disruption of this pathway by the A77V mutation provides a mechanistic explanation for the observed calcium leak and arrhythmogenic phenotype associated with the mutation. The identification of E171/E173 as potential calcium‑binding sites opens avenues for targeted drug design or mutagenesis studies aimed at restoring proper energy flow.
Overall, the study demonstrates that (1) a coarse‑grained elastic network can reveal functional energy conduits in large ion channels, (2) these conduits coincide with conserved structural elements (Mir domains), (3) pathogenic point mutations can sever the conduits, leading to dysregulated calcium handling, and (4) insights from related receptors (IP3R) can guide hypotheses about ligand‑binding sites in RyR2. Future work may focus on refining the model with anharmonic terms, exploring other disease‑linked mutations, and leveraging the identified pathway for therapeutic intervention.
Comments & Academic Discussion
Loading comments...
Leave a Comment