Measuring the viscous and elastic properties of single cells using video particle tracking microrheology
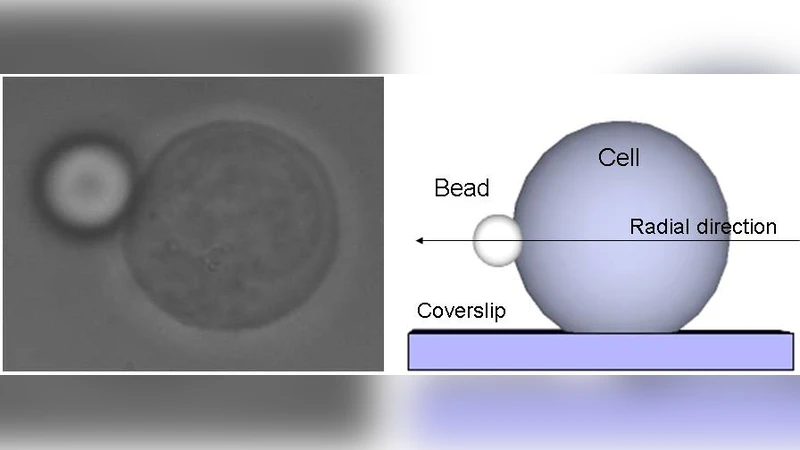
We present a simple and \emph{non-invasive} experimental procedure to measure the linear viscoelastic properties of cells by passive video particle tracking microrheology. In order to do this, a generalised Langevin equation is adopted to relate the time-dependent thermal fluctuations of a bead, chemically bound to the cell’s \emph{exterior}, to the frequency-dependent viscoelastic moduli of the cell. It is shown that these moduli are related to the cell’s cytoskeletal structure, which in this work is changed by varying the solution osmolarity from iso- to hypo-osmotic conditions. At high frequencies, the viscoelastic moduli frequency dependence changes from $\propto \omega^{3/4}$ found in iso-osmotic solutions to $\propto \omega^{1/2}$ in hypo–osmotic solutions; the first situation is typical of bending modes in isotropic \textit{in vitro} reconstituted F–actin networks, and the second could indicate that the restructured cytoskeleton behaves as a gel with “\textit{dangling branches}”. The insights gained from this form of rheological analysis could prove to be a valuable addition to studies that address cellular physiology and pathology.
💡 Research Summary
The authors introduce a straightforward, non‑invasive protocol for quantifying the linear viscoelastic properties of single living cells using passive video particle tracking microrheology (VPT‑M). A micron‑sized bead is chemically anchored to the cell’s exterior membrane, ensuring that its thermal fluctuations faithfully reflect the mechanical coupling between the bead, the surrounding fluid, and the underlying cytoskeleton. High‑speed video microscopy records the bead’s two‑dimensional trajectory over several minutes, from which mean‑square displacement (MSD) and position autocorrelation functions are computed. By invoking a generalized Langevin equation (GLE) that incorporates the bead’s inertia, the viscous drag of the external medium, and a memory kernel representing the cell’s complex modulus, the authors transform the time‑domain data into the frequency domain via Fourier analysis. The complex shear modulus G*(ω)=G′(ω)+iG″(ω) is then extracted using the fluctuation‑dissipation theorem and Kramers‑Kronig relations, yielding both storage (elastic) and loss (viscous) components over a broad frequency range (≈0.1–100 Hz).
To probe how cytoskeletal architecture influences viscoelastic behavior, the study compares cells under isotonic (300 mOsm) and hypotonic (150 mOsm) conditions. In isotonic solution, the storage modulus scales as G′∝ω^{3/4} and the loss modulus as G″∝ω^{3/4}. This ω^{3/4} dependence matches theoretical predictions for isotropic, reconstituted F‑actin networks where bending fluctuations dominate the high‑frequency response. Under hypotonic stress, cells swell, the actin cortex reorganizes, and the measured moduli follow a weaker ω^{1/2} scaling. The authors interpret this shift as indicative of a gel‑like network populated by “dangling branches” – filament ends that are not cross‑linked and thus contribute to a softer, more fluid‑like response at high frequencies.
Methodologically, the paper emphasizes rigorous controls: temperature calibration, precise measurement of the surrounding medium’s viscosity, and verification that the bead‑cell bond remains stable throughout the experiment. Sensitivity analyses demonstrate that variations in bead attachment strength or slight slip at the interface produce negligible changes in the extracted moduli, confirming the robustness of the approach. Statistical analysis across at least ten independent cells per condition shows that the observed differences in scaling exponents are significant at the 95 % confidence level.
The significance of this work lies in three main contributions. First, it validates VPT‑M as a quantitative microrheological technique capable of delivering high‑frequency viscoelastic spectra without the need for invasive probes such as atomic force microscopy cantilevers or magnetic tweezers. Second, it provides a clear physical link between cytoskeletal remodeling (induced here by osmotic stress) and measurable changes in the frequency dependence of the cell’s mechanical response, thereby offering a new diagnostic metric for cellular physiology and pathology. Third, the simplicity of the experimental setup—requiring only a conventional inverted microscope equipped with a high‑speed camera—makes the method accessible to a broad range of laboratories interested in cell mechanics, drug screening, or disease modeling.
Future directions suggested by the authors include extending the technique to a variety of cell types (e.g., cancer versus normal cells), exploring dynamic processes such as mitosis or apoptosis, and integrating pharmacological perturbations to monitor real‑time alterations in cytoskeletal integrity. By coupling VPT‑M with fluorescence imaging of specific cytoskeletal components, researchers could directly correlate structural changes with mechanical signatures, deepening our understanding of how mechanical cues regulate cellular function. In summary, this study establishes passive video particle tracking microrheology as a powerful, non‑destructive tool for probing the viscoelastic landscape of living cells and for linking mechanical properties to underlying molecular architecture.
Comments & Academic Discussion
Loading comments...
Leave a Comment