Asymptotic Analysis of Microtubule-Based Transport by Multiple Identical Molecular Motors
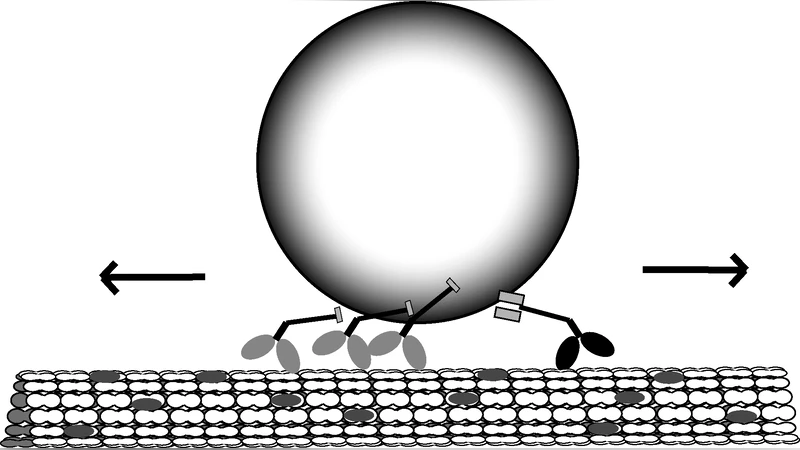
We describe a system of stochastic differential equations (SDEs) which model the interaction between processive molecular motors, such as kinesin and dynein, and the biomolecular cargo they tow as part of microtubule-based intracellular transport. We show that the classical experimental environment fits within a parameter regime which is qualitatively distinct from conditions one expects to find in living cells. Through an asymptotic analysis of our system of SDEs, we develop a means for applying in vitro observations of the nonlinear response by motors to forces induced on the attached cargo to make analytical predictions for two parameter regimes that have thus far eluded direct experimental observation: 1) highly viscous in vivo transport and 2) dynamics when multiple identical motors are attached to the cargo and microtubule.
💡 Research Summary
The paper presents a mathematically rigorous framework for describing intracellular transport driven by multiple identical processive molecular motors (e.g., kinesin‑1) pulling a cargo along a microtubule. Starting from a system of stochastic differential equations (SDEs), the authors model each motor’s position (X_i(t)) and the cargo’s position (Z(t)) as continuous variables subject to force‑dependent drift and diffusion. The motor drift is given by a scaled velocity function (v,g(F/F^)) where (F) is the elastic force generated by the motor‑cargo linker, (F^) is the motor stall force, and (g) captures the experimentally measured force‑velocity relationship. The diffusion term (\sigma,h(F/F^*)) represents stochastic stepping; the authors set (h\equiv1) for simplicity. The cargo obeys an overdamped Langevin equation with Stokes drag (\gamma=6\pi a\eta) and thermal noise (\sqrt{2k_BT\gamma}), plus any external laser‑trap force (\theta).
A nondimensional analysis identifies two small parameters: (\varepsilon = k_BT/(F^L)) (thermal to stall‑force ratio) and (\delta = \gamma v/F^) (drag‑to‑stall‑force ratio). In typical in‑vitro experiments (water‑like viscosity), (\delta\ll1); in vivo cytoplasm, with viscosity up to 600× water, (\delta) can approach or exceed unity. This separation of scales enables stochastic averaging. When (\delta\ll1), cargo dynamics are fast; the cargo quickly reaches a quasi‑stationary distribution conditioned on the slower motor positions. Averaging over this distribution yields an effective drift for each motor (equation (8)), showing that the overall transport velocity becomes essentially independent of the fluid viscosity—a phenomenon observed experimentally by Gross et al. and reproduced in silico by Kunwar & Mogilner.
In the high‑viscosity regime ((\delta\gtrsim1)), cargo motion is slow and the elastic linker is stretched. The authors derive a velocity formula (13) that explicitly depends on the linker spring constant (k); increasing (k) reduces the cargo‑motor complex speed. By equating the drag force at high viscosity with an externally applied force in low‑viscosity assays, they obtain a general conversion (equation (16)) and its inverse (equation (18)), providing a “back‑of‑the‑envelope” tool for translating in‑vitro measurements to in‑vivo predictions. Their analysis predicts that stall force should increase with viscosity, contrary to some experimental reports (Shubeita et al.), suggesting that additional mechanisms (e.g., near‑stall motor conformational changes) must be incorporated.
The paper then focuses on the case of two identical motors (N=2). Two reduced models are examined. The first, called the “instant relaxation” model, assumes the cargo instantaneously sits at the midpoint between the motors, effectively making cargo dynamics infinitely fast. Under this assumption, the relative motor separation (Y = X_2 - X_1) follows a one‑dimensional SDE whose drift depends on the shape of the single‑motor force‑velocity curve. The authors prove Theorem 5.1: whether two motors move faster or slower than a single motor hinges on the curvature of the force‑velocity relation; a sharply saturating curve can make the two‑motor system slower. They also show that the stall force of the two‑motor system exceeds twice the single‑motor stall force, a super‑additive effect (Proposition 5.2). This contrasts with earlier models that reported sub‑additivity, implying that the previously observed sub‑additivity may stem from motor binding/unbinding dynamics absent in the present analysis.
Throughout, the authors compare their analytical results with existing literature, highlighting where their more detailed stochastic treatment (including motor diffusion) alters predictions. They acknowledge limitations: the model neglects motor attachment/detachment, assumes a linear elastic linker, and treats the cytoplasm as a Newtonian fluid rather than a viscoelastic medium. They propose future work to incorporate binding kinetics, nonlinear linker mechanics, and viscoelastic drag, which should reconcile the discrepancy regarding stall‑force dependence on viscosity.
In summary, the paper delivers a clear asymptotic framework that bridges low‑viscosity in‑vitro experiments and high‑viscosity in‑vivo conditions, provides explicit formulas for transport velocity and stall force under varying viscosities, and elucidates how multiple identical motors interact through the cargo. The analytical insights, especially the identification of parameter regimes via (\delta) and the super‑additive stall force result, offer valuable guidance for interpreting existing data and designing new experiments probing multi‑motor transport in realistic cellular environments.
Comments & Academic Discussion
Loading comments...
Leave a Comment