Identification of network motifs capable of frequency-tunable and robust oscillation
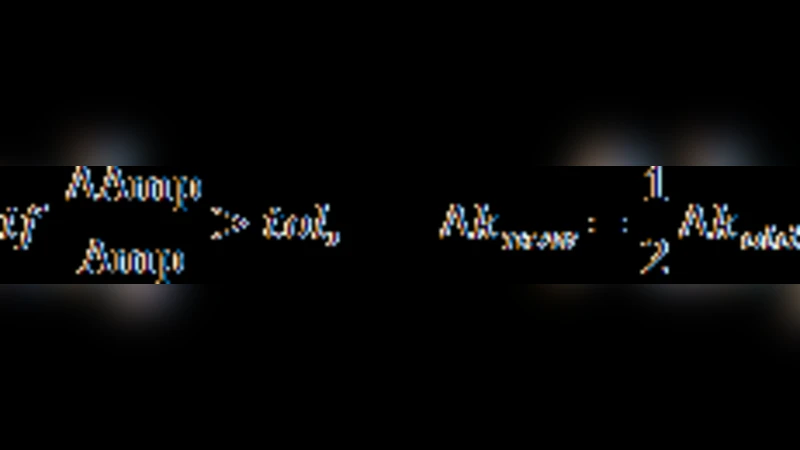
Oscillation has an important role in bio-dynamical systems such as circadian rhythms and eukaryotic cell cycle. John Tyson et. al. in Nature Review Mol Cell Biol 2008 examined a limited number of network topologies consisting of three nodes and four or fewer edges and identified the network design principles of biochemical oscillations. Tsai et. al. in Science 2008 studied three different network motifs, namely a negative feedback loop, coupled negative feedback loops, and coupled positive and negative feedback loops, and found that the interconnected positive and negative feedback loops are capable of generating frequency-tunable oscillations. We enumerate 249 topologically unique network architectures consisting of three nodes and at least three cyclic inhibitory edges, and identify network architectural commonalities among three functional groups: (1) most frequency-tunable yet less robust oscillators, (2) least frequency-tunable and least robust oscillators, and (3) less frequency-tunable yet most robust oscillators. We find that Frequency-tunable networks cannot simultaneously express high robustness, indicating a tradeoff between frequency tunability and robustness.
💡 Research Summary
**
The paper investigates how network architecture influences two key properties of biochemical oscillators: frequency tunability (the ability to vary the oscillation frequency over a wide range without large changes in amplitude) and robustness (the ability to maintain oscillations under parameter perturbations). Building on earlier work that examined a limited set of three‑node motifs, the authors systematically generate all topologically distinct three‑node networks that contain at least three inhibitory (negative) edges, arriving at a total of 249 unique architectures. The “backbone” of each network is a cyclic inhibitory loop (A → B → C → A). Additional edges are classified as auto‑regulatory (self‑loops) or para‑regulatory (inter‑node connections) and can be activating (+1), inhibiting (‑1), or absent (0). By accounting for symmetry, duplicate dynamical behaviors are eliminated, ensuring each of the 249 networks yields a distinct set of ordinary differential equations (ODEs).
Each network is modeled with first‑order ODEs using Michaelis–Menten/Hill kinetics for transcriptional regulation. The number of kinetic parameters varies from 12 to 45 depending on the presence of auto‑ and para‑regulatory links. Parameter space is explored via Monte‑Carlo sampling: for each network 1,000 random parameter sets are generated uniformly on a logarithmic scale. Numerical integration (fixed‑step RK4) is performed until a steady state or sustained oscillation is observed. A trajectory is classified as oscillatory if the sum of squared time‑derivatives remains above a small threshold (0.001) for a sufficiently long simulation window.
For each oscillatory parameter set, the authors locate the boundaries of the oscillatory domain by varying the synthesis/degradation rate constant (k) of node B using a binary search. The search continues until the upper and lower bifurcation points are resolved within 10⁻⁴. Within this domain, k is stepped adaptively to generate ~100 evenly spaced points; for each point the oscillation frequency, amplitude (max‑min of each node), and limit‑cycle distance are recorded. The “operational frequency range” is defined as the interval of k over which the amplitude varies by less than 50 %; the corresponding k‑range is the “operational k range.”
To quantify the two performance metrics, the authors compute the geometric mean of the operational frequency range and of the operational k range across the 1,000 parameter sets for each network. Networks are then plotted in a two‑dimensional space (frequency tunability vs. robustness) and clustered into three functional groups:
-
Group I (high robustness) – Networks featuring two auto‑positive feedback loops linked by a para‑negative feedback. These exhibit large operational k ranges, narrow frequency ranges, and predominantly subcritical Hopf bifurcations. Sensitivity analysis shows they are resilient to variations in auto‑positive feedback strength but vulnerable to weakening of the backbone negative loop.
-
Group IV (high frequency tunability) – Networks with two auto‑positive feedback loops linked by a para‑positive feedback. They display the widest operational frequency ranges, broad k ranges, and mainly supercritical Hopf bifurcations, indicating lower robustness. Perturbations of the backbone inhibitory strength quickly extinguish oscillations.
-
Group II (low tunability, low robustness) – Networks with two auto‑negative feedback loops linked by a para‑positive feedback. Both tunability and robustness are modest, and oscillations are often confined to a narrow parameter window.
Bifurcation diagrams for representative networks illustrate that high‑robustness motifs cluster near the subcritical Hopf region, whereas high‑tunability motifs cluster near the supercritical Hopf region, confirming an inherent trade‑off: a network cannot simultaneously achieve maximal frequency tunability and maximal robustness.
The authors also perform a local sensitivity analysis, perturbing each kinetic parameter by ±10 %. Results indicate that auto‑positive feedback strengths have a relatively mild impact on oscillation persistence, whereas the strength of the backbone inhibitory loop is a critical determinant of both frequency range and robustness.
In conclusion, the study provides a comprehensive map linking three‑node network topology to two essential dynamical properties of biochemical oscillators. The identified trade‑off between frequency tunability and robustness offers a design principle for synthetic biology: to obtain a highly tunable clock, one should employ motifs with coupled positive feedbacks; to build a robust clock, motifs with coupled negative feedbacks are preferable. This framework can guide the engineering of synthetic gene circuits and deepen our understanding of how natural regulatory networks balance flexibility and stability.
Comments & Academic Discussion
Loading comments...
Leave a Comment