Molecular Labor Division: Its Cause and Consequence
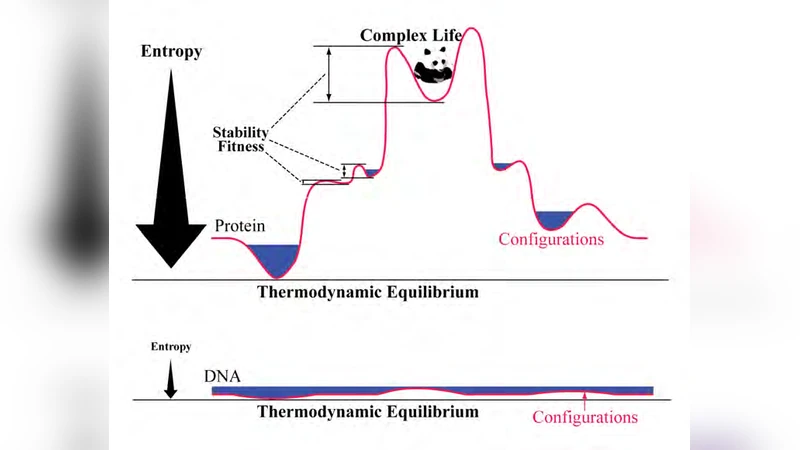
Both external environmental selection and internal lower-level evolution are essential for an integral picture of evolution. This paper proposes that the division of internal evolution into DNA/RNA pattern formation (genotype) and protein functional action (phenotype) resolves a universal conflict between fitness and evolvability. Specifically, this paper explains how this universal conflict drove the emergence of genotype-phenotype division, why this labor division is responsible for the extraordinary complexity of life, and how the specific ways of genotype-phenotype mapping in the labor division determine the paths and forms of evolution and development.
💡 Research Summary
The paper tackles a long‑standing paradox in evolutionary biology: how organisms can simultaneously maximize current fitness while retaining the capacity to generate future adaptations. The author defines this as the “fitness‑evolvability conflict.” High fitness usually requires a stable, well‑adapted phenotype, which in turn limits the raw material for evolutionary change because fewer beneficial mutations can be tolerated. Conversely, a high mutation rate supplies the raw material for future adaptation but tends to reduce present‑day fitness. To resolve this dilemma, the author proposes a universal “molecular labor division” between the genome (DNA/RNA) and the proteome (proteins). The genome’s primary role is pattern formation: it stores, replicates, and mutates information. Because replication and mutation are intrinsic to nucleic acids, the genome can sustain a relatively high mutation rate without immediate loss of function. Proteins, on the other hand, are the functional executors. Their three‑dimensional structures and biochemical activities have been refined by deep evolutionary history, making them comparatively robust to random sequence changes. This separation allows a system in which the information‑bearing layer can explore sequence space freely while the functional layer provides phenotypic stability.
A central claim is that the specific ways in which genotype maps onto phenotype—through transcription factors, regulatory RNAs, epigenetic marks, and post‑translational modifications—shape the “evolutionary landscape.” These mapping mechanisms confer two key properties: modularity and reusability. Modularity means that a single genetic module (e.g., a HOX gene cluster) can be deployed in multiple tissues or developmental stages, enabling complex body plans without requiring a proportionate increase in genetic material. Reusability allows new functions to arise by recombining existing modules rather than inventing them de novo; enzyme families illustrate this by preserving a catalytic core while varying peripheral residues to acquire new substrate specificities. The paper supports these ideas with empirical evidence: (1) engineered gene rearrangements in E. coli that quickly generate novel metabolic pathways, (2) directed‑evolution experiments on enzymes that demonstrate rapid functional diversification, and (3) comparative genomics showing conserved protein domains paired with divergent regulatory sequences across metazoans.
The author also examines systems that lack a clear labor division, such as RNA viruses where the genome itself performs catalytic functions. In these cases, the fitness‑evolvability conflict becomes acute: high mutation rates can quickly erode functional capacity, leading to frequent population crashes or limited adaptive potential. This contrast underscores the evolutionary advantage conferred by separating information storage from functional execution.
Finally, the paper outlines two broader implications. First, the genotype‑phenotype mapping constrains the set of viable evolutionary trajectories, effectively defining a “potential space” that determines which innovations are reachable. Second, synthetic biology can harness the labor‑division principle: by designing stable protein scaffolds coupled with highly mutable nucleic‑acid‑based control circuits, engineers can create systems that retain functional robustness while exploring vast design spaces. In sum, the molecular labor division between DNA/RNA and proteins is presented as the fundamental mechanism that reconciles the opposing demands of fitness and evolvability, enabling the extraordinary complexity observed in living systems and providing a powerful conceptual framework for both evolutionary theory and bio‑engineering.
Comments & Academic Discussion
Loading comments...
Leave a Comment