Energy conversion in Purple Bacteria Photosynthesis
The study of how photosynthetic organisms convert light offers insight not only into nature’s evolutionary process, but may also give clues as to how best to design and manipulate artificial photosynthetic systems – and also how far we can drive natural photosynthetic systems beyond normal operating conditions, so that they can harvest energy for us under otherwise extreme conditions. In addition to its interest from a basic scientific perspective, therefore, the goal to develop a deep quantitative understanding of photosynthesis offers the potential payoff of enhancing our current arsenal of alternative energy sources for the future. In the following Chapter, we consider the trade-off between dynamics, structure and function of light harvesting membranes in Rps. Photometricum purple bacteria, as a model to highlight the priorities that arise when photosynthetic organisms adapt to deal with the ever-changing natural environment conditions.
💡 Research Summary
The paper presents a comprehensive quantitative study of the energy conversion processes in the photosynthetic membranes of the purple bacterium Rps. photometricum. It begins by emphasizing the dual scientific relevance of understanding natural photosynthesis: gaining insight into evolutionary optimization and informing the design of artificial photosynthetic devices. The authors focus on how the bacterium adapts its light‑harvesting architecture under varying illumination conditions, specifically low‑light intensity (LLI, ~10 W m⁻²) and high‑light intensity (HLI, ~100 W m⁻²) environments that are typical of its natural habitats in dimly lit aquatic zones.
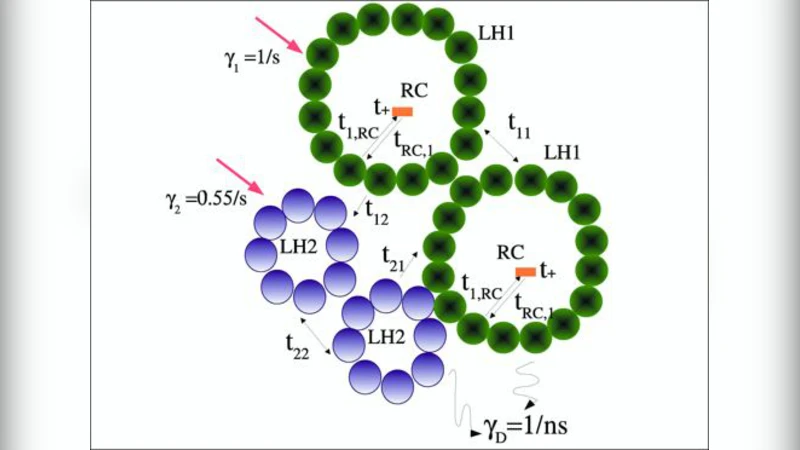
The structural overview details the two main light‑harvesting complexes, LH1 and LH2. LH2 consists of nine αβ subunits, each containing a single B800 bacteriochlorophyll (BChl) and a pair of B850 BChl pigments, plus one carotenoid per subunit. LH1 contains sixteen αβ units (32 BChl) that surround a reaction center (RC). The authors provide absorption cross‑section calculations, yielding photon capture rates of roughly 1 s⁻¹·W⁻¹·m² for LH1 and 0.55 s⁻¹·W⁻¹·m² for LH2. For a vesicle containing N₁ LH1 and N₂ LH2 complexes, the total absorption rate scales as I(γ₁N₁ + γ₂N₂), where I is the incident light intensity.
Excitation energy transfer is modeled using a combination of quantum‑mechanical Coulomb coupling and classical Förster theory. Within a single complex, strong dipole‑dipole interactions (e.g., 806 cm⁻¹ for nearest‑neighbor B850 pigments) generate delocalized excitonic states that enable sub‑picosecond transfer. The weaker coupling in the B800 ring (≈377 cm⁻¹) leads to rapid vibrational thermalization, which actually facilitates robust B800→B850 transfer. Inter‑complex transfer, occurring over 20–100 Å, is treated as an incoherent rate process. The authors derive the Förster rate k_DA = (2π/ħ)|U_DA|²∫G_D(E)G_A(E)dE, where G_D and G_A are Franck‑Condon weighted densities of states obtained from measured absorption and fluorescence spectra. Using published spectroscopic data, they reproduce experimentally observed transfer times: LH2→LH1 ≈ 3.3 ps, LH1→LH2 ≈ 15.5 ps, and estimate LH1→LH1 ≈ 20 ps. LH1→RC transfer is also discussed in terms of symmetry‑breaking of the B850 ring.
To capture the stochastic nature of exciton migration across many complexes, the authors implement a classical random‑walk model. Each step corresponds to an inter‑complex transfer event with probabilities derived from the Förster rates. The walk is coupled to the downstream electron transport chain (RC → cytochrome bc₁ → quinone Q_B → RC) and to proton translocation that drives ATP synthase. By varying N₁/N₂ ratios, they simulate vesicles under LLI and HLI conditions. Atomic force microscopy (AFM) observations show that LLI vesicles are larger, contain more LH2 complexes, and thus have a larger absorption cross‑section but longer electron‑transport pathways. HLI vesicles are more compact, enriched in LH1, yielding shorter pathways and higher conversion efficiency. The model predicts that the overall quantum efficiency (photons absorbed → ATP produced) can shift from ~5 % under LLI to ~12 % under HLI, reflecting the structural re‑organization.
The paper also explores extreme illumination scenarios, such as ultra‑high photon fluxes or non‑Poissonian photon arrival statistics that might be encountered in space or during catastrophic solar events. Simulations reveal a saturation point where the electron transport chain becomes bottlenecked, the proton motive force collapses, and ATP synthesis efficiency drops sharply. This defines a “survivability margin” beyond which the bacteria cannot maintain metabolic function. The authors suggest that such margins are crucial for assessing the feasibility of using engineered photosynthetic microbes in extraterrestrial environments or for bio‑hybrid energy systems.
In conclusion, the study demonstrates that global structural changes in the chromatophore membrane—driven by light intensity—play a more decisive role in overall energy conversion than the detailed quantum coherence within individual complexes. By integrating high‑resolution structural data, spectroscopic measurements, and stochastic transport modeling, the authors provide a quantitative framework that links membrane architecture, excitation dynamics, and metabolic output. This framework not only deepens our understanding of purple bacterial photosynthesis but also offers concrete design principles for artificial light‑harvesting assemblies and for engineering robust, microbe‑based energy production under diverse and extreme environmental conditions.
Comments & Academic Discussion
Loading comments...
Leave a Comment