Magnetically-triggered Nanocomposite Membranes: a Versatile Platform for Triggered Drug Release
Drug delivery devices based on nanocomposite membranes containing thermoresponsive nanogels and superparamagnetic nanoparticles have been demonstrated to provide reversible, on-off drug release upon application (and removal) of an oscillating magnetic field. The dose of drug delivered can be tuned by engineering the phase transition temperature of the nanogel, the loading of nanogels in the membrane, and the membrane thickness, allowing for the delivery of drugs over several orders of magnitude of release rates. The zero-order kinetics of drug release through the membranes permit drug doses from a specific device to be tuned according to the duration of the magnetic field. Drugs over a broad range of molecular weights (500-40,000 Da) can be delivered by the same membrane device. Membrane-to-membrane and cycle-to-cycle reproducibility is demonstrated, suggesting the general utility of these membranes for drug delivery.
💡 Research Summary
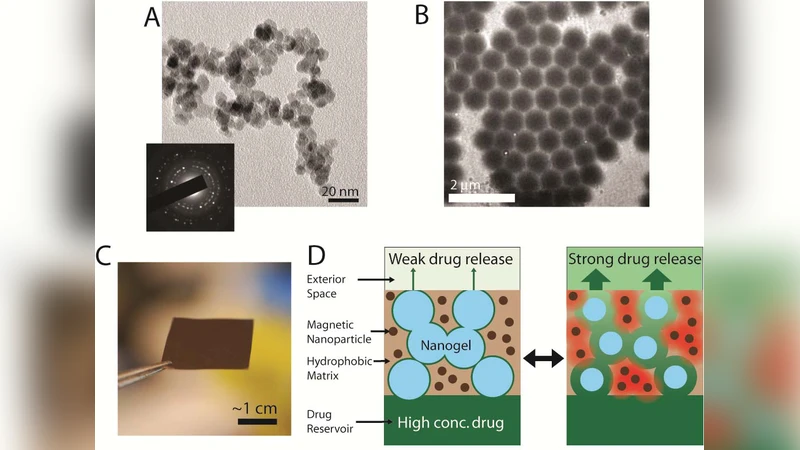
The authors present a magnetically‑triggered nanocomposite membrane platform that enables on‑demand, zero‑order drug release with tunable rates spanning several orders of magnitude. The membrane is fabricated by dispersing super‑paramagnetic iron oxide nanoparticles and temperature‑responsive poly(N‑isopropylacrylamide) (PNIPAm) based nanogels within an ethyl‑cellulose matrix, followed by solvent evaporation to form a thin film. By copolymerizing N‑isopropylmethacrylamide (NIPMAm) and acrylamide (AAm) with NIPAm, the nanogels’ volume‑phase transition temperature (VPTT) can be shifted from ~32 °C to ~46 °C without altering the magnitude of swelling/deswelling. When an alternating magnetic field is applied, the magnetic nanoparticles generate localized heating, causing the nanogels to collapse. This collapse opens a disordered pore network throughout the membrane, dramatically increasing its permeability.
Key design variables are (i) nanogel VPTT, (ii) nanogel loading (wt % in the membrane), and (iii) membrane thickness. VPTT determines the temperature at which the membrane switches from the low‑permeability “off” state to the high‑permeability “on” state; a modest offset (~2 °C) between nanogel VPTT and flux transition is observed due to the need for sufficient free volume generation. Increasing membrane thickness linearly reduces the steady‑state flux (R² = 0.92), providing a straightforward method to set a baseline release rate. Nanogel loading exerts a logarithmic influence on flux: 12–32 wt % loading raises the on‑state flux by up to two orders of magnitude, while simultaneously decreasing the on/off flux ratio (e.g., from 15.2 ± 2.6 at 12 wt % to 6.0 ± 0.5 at 32 wt %). Thus, an optimal loading window (≈20–30 wt %) balances high flux with sufficient on/off contrast.
All configurations exhibit zero‑order kinetics for at least 24 h in both on and off states (R² > 0.98). Consequently, the total drug dose delivered can be precisely programmed by the duration of magnetic field exposure, independent of the intrinsic release rate. The platform accommodates a broad spectrum of therapeutics: small molecules (≈500 Da sodium fluorescein), larger macromolecules (40 kDa FITC‑dextran), and amphiphilic cationic drugs (bupivacaine) all demonstrate on/off switching with comparable flux ratios, indicating that the membrane’s physical properties dominate release behavior rather than drug chemistry.
Reproducibility is high. Cycle‑to‑cycle variability for a single membrane over four on/off cycles is statistically insignificant (p > 0.07). Device‑to‑device variability is minimal for high‑loading membranes (p > 0.18) but modestly higher for low‑loading membranes, likely due to manual mixing inconsistencies; automation could mitigate this.
In summary, the study establishes a versatile, magnetically‑actuated nanocomposite membrane that can be rationally engineered via nanogel composition, loading, and thickness to achieve programmable, on‑demand drug delivery with zero‑order kinetics, broad molecular weight applicability, and excellent reproducibility. This platform offers a promising route toward patient‑specific, remotely controlled therapeutics for chronic diseases requiring precise dosing such as diabetes, pain management, or cancer therapy.
Comments & Academic Discussion
Loading comments...
Leave a Comment