Functional modularity of background activities in normal and epileptic brain networks
We analyze the connectivity structure of weighted brain networks extracted from spontaneous magnetoencephalographic (MEG) signals of healthy subjects and epileptic patients (suffering from absence seizures) recorded at rest. We find that, for the activities in the 5-14 Hz range, healthy brains exhibit a sparse connectivity, whereas the brain networks of patients display a rich connectivity with clear modular structure. Our results suggest that modularity plays a key role in the functional organization of brain areas during normal and pathological neural activities at rest.
💡 Research Summary
In this study the authors investigated how the functional connectivity of the brain differs between healthy individuals and patients with absence epilepsy during a resting, non‑seizure state. Magnetoencephalographic (MEG) recordings were obtained from five control subjects and five patients, each measured with a whole‑head 151‑sensor system at a sampling rate of 1.25 kHz. From each recording, 338 artefact‑free, 5‑second quasi‑stationary segments were extracted (206 from patients, 132 from controls). The focus was on the extended alpha band (5–14 Hz), because only in this frequency range did the two groups show statistically significant differences.
Functional links between every pair of sensors were quantified using linear coherence. Power and cross‑spectral densities were estimated with Welch’s averaged periodogram, and the squared magnitude of coherence |Γij(f)|² was computed. To reduce spurious short‑range correlations, sensors closer than 5 cm were excluded. Fisher’s Z‑transform was applied to the coherence values, yielding normally distributed Zij under the null hypothesis of independence. Multiple‑comparison correction was performed with the False Discovery Rate (q ≤ 0.01); only connections exceeding the resulting threshold Γth were retained, and their weights were set to the original coherence value, producing an undirected weighted graph of N = 151 nodes.
A suite of weighted‑graph metrics was then calculated: node strength si = ∑j wij, weighted clustering coefficient ci, nodal efficiency Ei, and their averages across the network (S, Cw, E). Randomized reference graphs were generated by rewiring edges while preserving the degree distribution. Both patient and control networks displayed higher clustering than random graphs (p < 10⁻³), confirming a small‑world organization. However, patients exhibited significantly larger average strength (S) and clustering (Cw) than controls (p < 10⁻³), indicating a denser functional connectivity in the epileptic brain even during inter‑ictal periods.
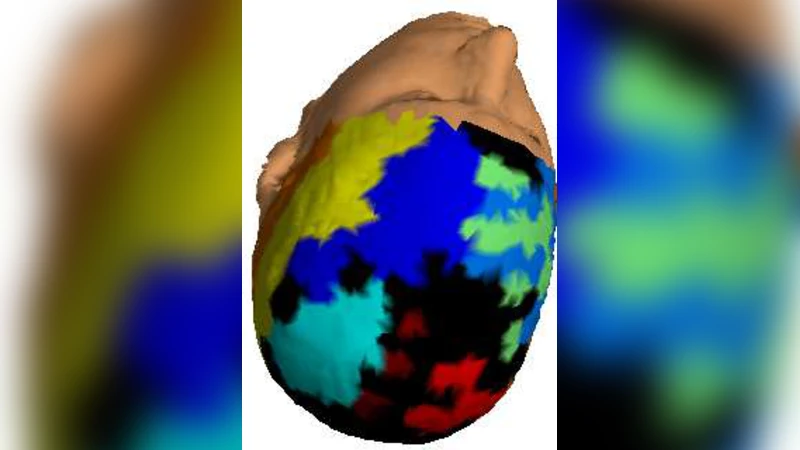
To uncover modular organization, the authors employed a diffusion‑map based spectral embedding. A Markov transition matrix P was constructed from the weighted adjacency matrix, and its eigenvalues λk and eigenvectors ψk were used to define a diffusion distance dij(t). By retaining the first β non‑trivial eigenvectors, each node was embedded into a low‑dimensional Euclidean space Rβ. k‑means clustering was then applied repeatedly (500 initializations) for candidate numbers of modules Nm = 2…15. For each partition the Newman‑type modularity Q was computed; the partition with maximal Q was selected as the optimal community structure. This procedure yielded, on average, 13.7 modules for controls and 8.3 modules for patients, with patients showing fewer but larger modules. The stability of the partitions was assessed with the Adjusted Rand Index (J), giving J = 0.733 for patients (high consistency) and J = 0.479 for controls (more variability).
Node‑level analyses focused on two complementary measures: the within‑module degree z‑score (zi) and the participation coefficient (pci). The z‑score quantifies how strongly a node connects to other nodes in its own module relative to the module’s average strength, while the participation coefficient measures the extent to which a node’s links are distributed across different modules. Controls displayed high zi values and low pci, reflecting strong intra‑module cohesion and limited inter‑module connectivity. In contrast, patients—particularly in the right centro‑parietal region—showed markedly elevated pci, indicating that many of their nodes act as bridges linking multiple modules. The distribution of zi differed only slightly between groups, suggesting that intra‑module connectivity strength is comparable, whereas the pattern of inter‑module integration diverges.
Statistical significance of all graph metrics was evaluated using Chebyshev’s inequality for deviation from random‑graph expectations and non‑parametric permutation testing (10⁵ permutations) to account for spatial dependencies. The authors also verified that differences were not driven merely by a higher number of edges, as random rewiring preserving degree eliminated the observed disparities in clustering and efficiency.
In summary, the paper demonstrates that resting‑state functional brain networks in the alpha band are structurally distinct in absence epilepsy. Epileptic brains exhibit richer connectivity, higher clustering, and a modular architecture composed of fewer, larger communities. Moreover, the altered participation patterns—especially the increased cross‑module linking in centro‑parietal areas—suggest a propensity for widespread integration that may facilitate the emergence of seizure activity. These findings support the view that modularity is a key organizational principle differentiating normal from pathological neural dynamics and propose modularity‑based metrics as potential biomarkers for epilepsy. Future work could extend this approach to pre‑ictal and post‑ictal states, assess treatment effects, and explore whether similar modular alterations are present in other neurological disorders.
Comments & Academic Discussion
Loading comments...
Leave a Comment