Polarity patterns of stress fibers
Stress fibers are contractile actomyosin bundles commonly observed in the cytoskeleton of metazoan cells. The spatial profile of the polarity of actin filaments inside contractile actomyosin bundles is either monotonic (graded) or periodic (alternating). In the framework of linear irreversible thermodynamics, we write the constitutive equations for a polar, active, elastic one-dimensional medium. An analysis of the resulting equations for the dynamics of polarity shows that the transition from graded to alternating polarity patterns is a nonequilibrium Lifshitz point. Active contractility is a necessary condition for the emergence of sarcomeric, alternating polarity patterns.
💡 Research Summary
The paper addresses a fundamental question in cell biology: why do actin‑myosin bundles (stress fibers) exhibit either a monotonic (graded) or a periodic (alternating) polarity of actin filaments, and what physical mechanisms control the transition between these two patterns. To answer this, the authors construct a continuum description of a one‑dimensional polar, active, elastic medium. The state variables are the displacement field u(x,t) describing bundle deformation and the polarity field p(x,t) that encodes the local orientation of actin filaments. Using linear irreversible thermodynamics, they write constitutive relations for the stress σ and the polarity flux Jp that include both passive (elastic, viscous, diffusive) and active contributions driven by ATP hydrolysis (Δμ). The stress reads
σ = G ∂x u + η ∂t ∂x u + ζ p + α Δμ,
where G is the elastic modulus, η the viscosity, ζ couples polarity to stress, and α Δμ is the active contractile stress generated by myosin motors. The polarity flux is
Jp = –D ∂x p + λ ∂x σ + β Δμ p,
with D the polarity diffusion coefficient, λ a stress‑gradient‑induced drift, and β a term that amplifies polarity in the presence of activity.
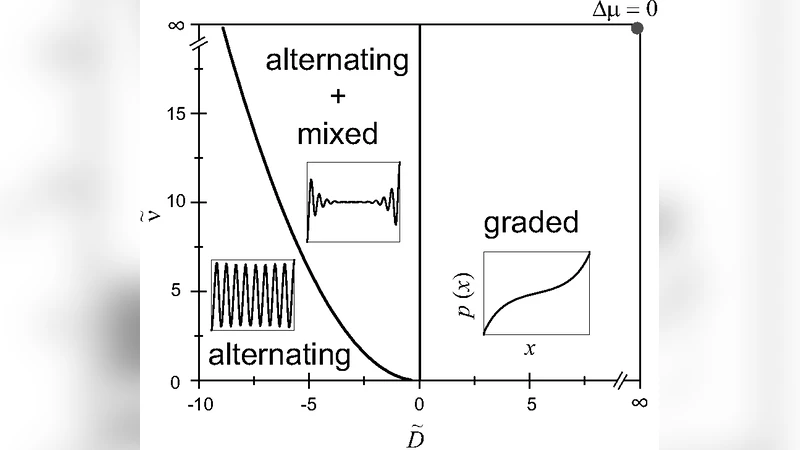
Combining force balance (∂x σ = 0) with the continuity equation for polarity (∂t p + ∂x Jp = 0) yields a coupled set of nonlinear partial differential equations. The authors first perform a linear stability analysis around the homogeneous state (p = 0, u = 0). Two distinct unstable modes emerge: a long‑wavelength mode (k → 0) that leads to a smooth, graded polarity profile, and a finite‑wavelength mode (k = kc) that generates a sinusoidal alternation of polarity. The control parameters are the active contractility α Δμ and the polarity diffusion D. When contractility is weak (α Δμ < αc Δμ) and diffusion is strong (D > Dc), only the graded mode is unstable, producing monotonic polarity. When contractility exceeds a critical value while diffusion is sufficiently low (α Δμ > αc Δμ, D < Dc), the finite‑wavelength mode dominates, giving rise to alternating polarity reminiscent of sarcomeric organization.
Crucially, the point at which the two regimes meet is identified as a nonequilibrium Lifshitz point. At this point the effective free‑energy‑like functional contains both k² and k⁴ terms, analogous to the Cahn‑Hilliard description of microphase separation, but here the “free energy” is maintained out of equilibrium by continuous ATP consumption. This theoretical insight explains why sarcomere‑like periodicity can only arise in actively contracting bundles: the energy input is required to stabilize the finite‑wavelength instability.
Numerical simulations of the full nonlinear equations confirm the analytical predictions. By scanning the (α Δμ, D) parameter plane the authors reproduce both experimentally observed patterns: graded polarity in cells with low myosin activity or high substrate stiffness, and alternating polarity in highly contractile cells (e.g., smooth muscle or fibroblasts under tension). The wavelength of the alternating pattern depends on the ratio of elastic to viscous coefficients and on the magnitude of the active stress, providing quantitative predictions that can be tested experimentally.
The paper’s conclusions have several important implications. First, they provide a unified physical framework that links molecular motor activity, filament polarity, and mechanical properties of the cytoskeleton. Second, they suggest that manipulating active contractility (e.g., through myosin inhibitors, ATP depletion, or optogenetic control of motor activity) or altering polarity diffusion (by changing cross‑linker density) can deliberately switch a cell’s stress‑fiber polarity from graded to alternating, offering a tool for cell‑shape engineering and tissue‑fabrication. Third, the identification of a nonequilibrium Lifshitz point extends concepts from equilibrium soft‑matter physics to living systems, highlighting how continuous energy consumption can generate and maintain ordered structures that would be impossible at thermodynamic equilibrium.
Finally, the authors outline future directions: extending the model to two‑ and three‑dimensional geometries, incorporating feedback between polarity and biochemical signaling pathways, and applying the framework to other polar filament assemblies such as microtubule bundles or actin cortex. Overall, the work bridges experimental observations of stress‑fiber polarity with a rigorous thermodynamic theory, revealing active contractility as the essential driver of sarcomeric, alternating polarity patterns.
Comments & Academic Discussion
Loading comments...
Leave a Comment