General analysis of mathematical models for bone remodeling
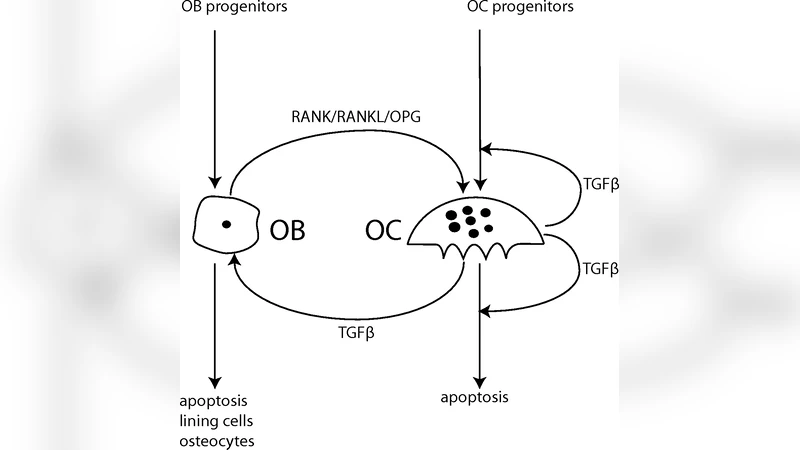
Bone remodeling is regulated by pathways controlling the interplay of osteoblasts and osteoclasts. In this work, we apply the method of generalized modelling to systematically analyse a large class of models of bone remodeling. Our analysis shows that osteoblast precursors can play an important role in the regulation of bone remodeling. Further, we find that the parameter regime most likely realized in nature lies very close to bifurcation lines, marking qualitative changes in the dynamics. Although proximity to a bifurcation facilitates adaptive responses to changing external conditions, it entails the danger of losing dynamical stability. Some evidence implicates such dynamical transitions as a potential mechanism leading to forms of Paget’s disease.
💡 Research Summary
This paper applies the framework of generalized modeling to a broad class of mathematical representations of bone remodeling, a process governed by the tightly coupled activities of osteoblasts (bone‑forming cells) and osteoclasts (bone‑resorbing cells). Rather than committing to specific functional forms for the rates of cell proliferation, differentiation, and death, the authors normalize the governing differential equations and replace each unknown function with its logarithmic derivative (elasticity) evaluated at the steady state. These elasticities serve as dimensionless parameters that capture the relative sensitivity of biological processes (e.g., RANKL/OPG signaling, hormonal regulation, mechanical loading) without requiring detailed kinetic specifications.
Through linear stability analysis of the steady‑state solutions, the study derives conditions under which the system exhibits Hopf or saddle‑node bifurcations. A key finding is that the elasticity associated with pre‑osteoblast proliferation and differentiation exerts a dominant influence on the location of the bifurcation surfaces. In other words, the precursor osteoblast compartment acts as a critical control node that can tip the balance between stable bone homeostasis and oscillatory or runaway remodeling.
Systematic exploration of the high‑dimensional elasticity space reveals that biologically realistic parameter sets lie remarkably close to bifurcation boundaries. This proximity implies that the skeletal system is poised to respond rapidly to external perturbations—such as changes in mechanical load or endocrine signals—by moving across a bifurcation and altering its dynamic regime. However, it also means that modest fluctuations in regulatory pathways can destabilize the system, leading to sustained oscillations or loss of the steady state.
The authors connect these theoretical insights to clinical observations of Paget’s disease, a disorder characterized by excessive and disorganized bone turnover. In the model, crossing a Hopf or saddle‑node bifurcation reproduces the pathological patterns seen in Paget’s disease, suggesting that the disease may arise from a shift of the regulatory elasticities beyond their critical thresholds, possibly due to genetic mutations (e.g., in SQSTM1) or aberrant cytokine signaling.
Finally, the paper highlights the practical utility of generalized modeling for guiding experiments and therapeutic design. By mapping elasticity sensitivities, researchers can identify which molecular targets (e.g., RANKL, OPG, sclerostin) are most effective for restoring stability, and can predict how pharmacological agents such as bisphosphonates or denosumab will move the system away from dangerous bifurcation zones. In sum, the work provides a rigorous, model‑agnostic framework that clarifies the central role of osteoblast precursors, explains why bone remodeling operates near critical points, and offers a mechanistic link between these dynamics and bone diseases like Paget’s.
Comments & Academic Discussion
Loading comments...
Leave a Comment