Multiscale Bone Remodelling with Spatial P Systems
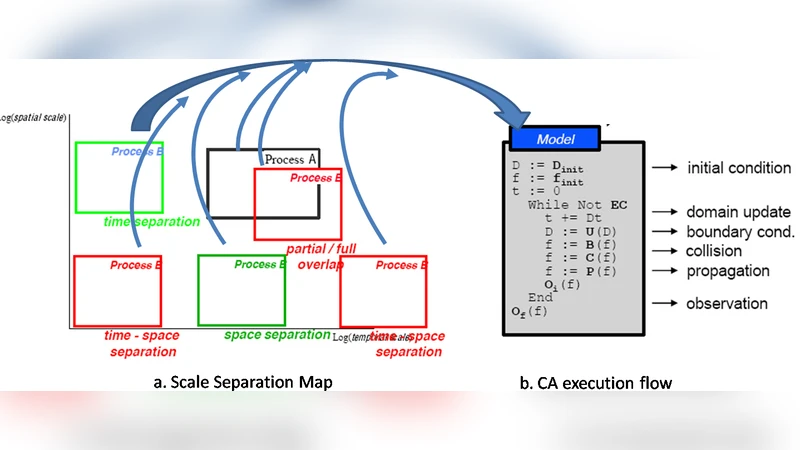
Many biological phenomena are inherently multiscale, i.e. they are characterized by interactions involving different spatial and temporal scales simultaneously. Though several approaches have been proposed to provide “multilayer” models, only Complex Automata, derived from Cellular Automata, naturally embed spatial information and realize multiscaling with well-established inter-scale integration schemas. Spatial P systems, a variant of P systems in which a more geometric concept of space has been added, have several characteristics in common with Cellular Automata. We propose such a formalism as a basis to rephrase the Complex Automata multiscaling approach and, in this perspective, provide a 2-scale Spatial P system describing bone remodelling. The proposed model not only results to be highly faithful and expressive in a multiscale scenario, but also highlights the need of a deep and formal expressiveness study involving Complex Automata, Spatial P systems and other promising multiscale approaches, such as our shape-based one already resulted to be highly faithful.
💡 Research Summary
The paper addresses the challenge of formally modelling biological processes that operate across multiple spatial and temporal scales, using bone remodeling as a concrete case study. It begins by reviewing existing multiscale approaches, highlighting that while several “multilayer” frameworks exist, only Complex Automata (CA)—an extension of Cellular Automata—naturally embed spatial information and provide well‑defined inter‑scale integration mechanisms. The authors then turn to Spatial P systems (SPS), a variant of membrane computing that augments traditional P systems with explicit geometric coordinates, allowing objects to occupy and move within a defined space.
A detailed comparison reveals that both CA and SPS share a hierarchical, layered architecture: a microscopic layer (cells, molecules) and a macroscopic layer (tissues, organs). In CA, inter‑scale coupling is achieved through upward and downward functions that aggregate microscopic events into macroscopic state variables and distribute macroscopic environmental cues back to the microscopic agents. SPS, however, implements these couplings via membrane boundaries and transition rules that can explicitly model material exchange, diffusion delays, and distance‑dependent interactions. This distinction gives SPS a finer‑grained expressive power while retaining the parallel, rule‑based dynamics characteristic of CA.
The core contribution is a two‑scale SPS model of bone remodeling. The microscopic layer consists of a three‑dimensional lattice populated by osteoclasts (bone‑resorbing cells) and osteoblasts (bone‑forming cells). Each cell type follows a set of transition rules governing activation, apoptosis, secretion of matrix, and response to biochemical signals (e.g., RANKL, OPG) and mechanical stimuli (e.g., strain). Diffusion of signaling molecules is captured by propagation rules that move objects to neighboring lattice sites, thereby reproducing spatial gradients. The macroscopic layer is a continuum mechanics description of bone tissue, providing stress‑strain fields and overall bone density.
Coupling between layers is realized through membrane boundaries that host two families of rules: (1) upward rules that aggregate the local resorption/formation rates into a change of macroscopic density, and (2) downward rules that translate macroscopic mechanical loads into adjustments of cellular proliferation and differentiation rates. The authors demonstrate that the SPS model reproduces the characteristic oscillatory dynamics of bone remodeling observed in both experimental data and previous CA models. Crucially, the SPS formulation captures heterogeneities such as localized overload, delayed signal propagation, and variable membrane permeability—features that are difficult to express in standard CA.
Simulation experiments include normal remodeling cycles, pathological conditions mimicking osteoporosis (increased osteoclast activity), and therapeutic interventions (anti‑resorptive drug delivery). In each scenario, the SPS model predicts spatially resolved outcomes (e.g., focal bone loss) that align with clinical observations, underscoring its higher fidelity.
The discussion emphasizes that while the SPS approach offers superior spatial expressiveness, a systematic study of formal expressiveness across multiscale frameworks is still lacking. The authors propose a meta‑theoretical analysis that would compare the operation sets, computational complexity, and verification capabilities of Complex Automata, Spatial P systems, and their previously developed shape‑based models. Such a study would lay the groundwork for a unified theory of multiscale modelling, facilitating model interoperability, validation, and eventual translation into biomedical engineering tools.
In conclusion, the paper successfully re‑frames the Complex Automata multiscale methodology within the Spatial P system formalism, delivers a biologically faithful two‑scale bone remodeling model, and highlights the need for deeper formal investigations to advance multiscale modelling across diverse scientific domains.
Comments & Academic Discussion
Loading comments...
Leave a Comment