The zipper mechanism in phagocytosis: energetic requirements and variability in phagocytic cup shape
Phagocytosis is the fundamental cellular process by which eukaryotic cells bind and engulf particles by their cell membrane. Particle engulfment involves particle recognition by cell-surface receptors, signaling and remodeling of the actin cytoskeleton to guide the membrane around the particle in a zipper-like fashion. Despite the signaling complexity, phagocytosis also depends strongly on biophysical parameters, such as particle shape, and the need for actin-driven force generation remains poorly understood. Here, we propose a novel, three-dimensional and stochastic biophysical model of phagocytosis, and study the engulfment of particles of various sizes and shapes, including spiral and rod-shaped particles reminiscent of bacteria. Highly curved shapes are not taken up, in line with recent experimental results. Furthermore, we surprisingly find that even without actin-driven force generation, engulfment proceeds in a large regime of parameter values, albeit more slowly and with highly variable phagocytic cups. We experimentally confirm these predictions using fibroblasts, transfected with immunoreceptor FcyRIIa for engulfment of immunoglobulin G-opsonized particles. Specifically, we compare the wild-type receptor with a mutant receptor, unable to signal to the actin cytoskeleton. Based on the reconstruction of phagocytic cups from imaging data, we indeed show that cells are able to engulf small particles even without support from biological actin-driven processes. This suggests that biochemical pathways render the evolutionary ancient process of phagocytic highly robust, allowing cells to engulf even very large particles. The particle-shape dependence of phagocytosis makes a systematic investigation of host-pathogen interactions and an efficient design of a vehicle for drug delivery possible.
💡 Research Summary
Phagocytosis, the ancient cellular process by which eukaryotic cells engulf particles, is traditionally viewed as a signaling‑driven event that relies heavily on actin‑mediated force generation. This paper challenges that view by constructing a three‑dimensional, stochastic biophysical model that treats the “zipper” mechanism—sequential receptor‑ligand binding that pulls the plasma membrane around a target—as a purely physical process, supplemented optionally by actin‑generated tension. The model discretizes particle surfaces into triangular meshes, distributes receptors at a defined density, and assigns a binding probability based on ligand affinity and thermal fluctuations. When a binding event occurs, the local membrane curvature is reduced, and the “zipper” propagates to neighboring mesh elements with a probability that depends on curvature, receptor density, and an optional actin term. By varying particle size, shape (spheres, rods, spirals), receptor density, and actin contribution, the authors map out regimes of successful engulfment, partial engulfment, and failure.
Key theoretical findings include: (1) Spherical particles between 0.5 µm and 5 µm can be fully engulfed even when the actin term is set to zero, although the engulfment rate drops by a factor of two to three compared with actin‑supported simulations. (2) Rod‑shaped particles are engulfed efficiently only when their length is short (<2 µm) or when actin provides additional contractile force; long rods (>10 µm) typically abort mid‑zipper in the absence of actin. (3) Highly curved or spiral geometries, which present a small effective binding area, rarely achieve a continuous zipper and are therefore not internalized. (4) Without actin, the phagocytic cup displays pronounced asymmetry and “incomplete zipper” regions, reflecting the mechanical instability of a membrane driven solely by stochastic binding.
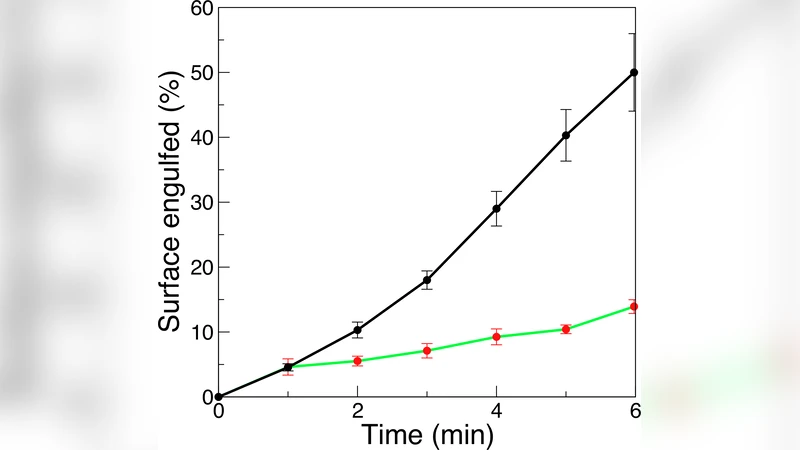
To validate the model, the authors engineered NIH‑3T3 fibroblasts to overexpress the human FcγRIIa receptor and generated a mutant lacking the ITAM motif that couples to the actin cytoskeleton. IgG‑opsonized particles of various shapes and sizes were presented to wild‑type (WT) and mutant cells. High‑resolution confocal microscopy combined with three‑dimensional reconstruction allowed quantitative measurement of cup morphology and engulfment success. WT cells engulfed >90 % of 0.5–5 µm spheres and >70 % of 3 µm × 10 µm rods, confirming the high efficiency predicted by the model when actin is present. Mutant cells, lacking actin signaling, still internalized ~30 % of 0.5 µm spheres but showed <10 % success for larger spheres, rods, and all spiral particles. The cups formed by mutants were irregular, often missing sections of membrane, exactly as the stochastic model predicted for an actin‑free scenario.
The discussion interprets these results in an evolutionary context. The ability of cells to engulf small particles without actin suggests that the core zipper mechanism is a robust, biophysical foundation that predates sophisticated signaling cascades. Actin, however, dramatically expands the size and shape envelope that can be handled, providing speed, symmetry, and mechanical stability—features essential for modern immune cells confronting diverse pathogens. The authors also highlight practical implications: because particle curvature and size dictate phagocytic outcome, rational design of drug‑delivery carriers can exploit these parameters to either promote uptake (e.g., low‑curvature, moderate‑size spheres) or avoid clearance (e.g., high‑curvature, elongated shapes). Conversely, understanding how pathogens might evolve shapes that evade the zipper mechanism could inform vaccine and antimicrobial strategies.
In summary, the study delivers a quantitative framework that integrates stochastic receptor‑ligand binding with optional actin‑driven tension, revealing that actin is not an absolute prerequisite for phagocytosis of small, low‑curvature particles but is essential for efficient engulfment of larger or more complex targets. This dual perspective—biophysical robustness plus biochemical amplification—advances our mechanistic grasp of phagocytosis and opens avenues for engineering both therapeutic particles and anti‑pathogen interventions.
Comments & Academic Discussion
Loading comments...
Leave a Comment