Towards Biosensing Strategies Based on Biochemical Logic Systems
Recent advances in biochemical computing, i.e., information processing with cascades of primarily enzymatic reactions realizing computing gates, such as AND, OR, etc., as well as progress in networking these gates and coupling of the resulting systems to smart/responsive electrodes for output readout, have opened new biosensing opportunities. Here we survey existing enabling research results, as well as ideas and research avenues for future development of a new paradigm of digitally operating biosensors logically processing multiple biochemical signals through Boolean logic networks composed of biomolecular reactions, yielding the final output signals as YES/NO responses. Such systems can lead to high-fidelity biosensing compared to common single or parallel sensing devices.
💡 Research Summary
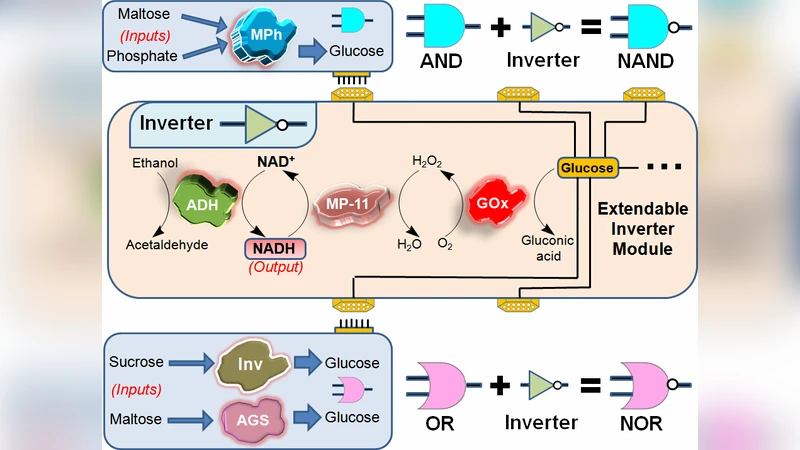
The paper presents a forward‑looking vision for “digital logic‑based biosensors,” a new class of analytical devices that process multiple biochemical inputs through enzymatic Boolean logic networks and deliver a binary (YES/NO) output via smart electrode read‑outs. The authors begin by reviewing the rapid progress made in biochemical computing over the past decade, focusing on the design of elementary logic gates—AND, OR, NOT, NAND, NOR—implemented with cascades of enzymatic reactions. By exploiting the kinetic parameters of enzymes (Km, Vmax) and the selective generation or consumption of small electroactive species (e.g., NADH, H₂O₂, acetate), each gate translates the presence or absence of specific analytes into a digital electrical signal.
Two principal strategies for coupling the enzymatic circuitry to an electrode are described. In the direct‑immobilization approach, enzymes are covalently or physically attached to conductive nanomaterials (carbon nanotubes, gold nanowires, graphene) so that electron transfer occurs over a minimal distance, yielding rapid and low‑noise signals. In the indirect or mediator‑based approach, the enzymatic reaction produces a diffusible electroactive product that is subsequently oxidized or reduced at the electrode, allowing signal amplification but introducing a diffusion delay. The choice between these architectures depends on the target application: high‑sensitivity clinical assays often favor mediator amplification, whereas point‑of‑care rapid tests benefit from the speed of direct electron transfer.
The paper then details how individual gates can be modularly assembled into multilayer logic networks. The output of one gate (e.g., a generated H₂O₂ pulse) serves as the input for the next, enabling the construction of complex Boolean functions such as (A AND B) OR (C NOT D). Critical design considerations include (1) reaction time constants—enzyme cascades typically operate on the scale of seconds to minutes, necessitating microfluidic flow control to reduce lag, (2) cross‑reactivity—orthogonal enzyme sets must be selected to avoid unintended signal mixing, and (3) electrode material optimization—high surface area, low charge‑transfer resistance substrates improve detection limits and dynamic range.
A comparative analysis demonstrates the advantages of logic‑based biosensing over conventional single‑analyte or parallel sensor arrays. For disease diagnostics that require the simultaneous presence of two biomarkers, an AND gate dramatically reduces false‑positive rates because a “positive” output is generated only when both markers exceed their thresholds. Conversely, an OR gate enhances sensitivity when either of several markers is sufficient for a diagnosis. NOT gates provide a means to suppress background interference from endogenous inhibitors, thereby improving signal‑to‑noise ratios. The authors quantify these benefits with simulated receiver‑operating‑characteristic (ROC) curves, showing that logical integration can shift the operating point toward higher specificity without sacrificing sensitivity.
Future research directions are outlined comprehensively. First, enzyme stability must be extended through immobilization in nanostructured matrices such as metal‑organic frameworks (MOFs) or hydrogel scaffolds, which also enable higher loading densities. Second, reaction latency can be minimized by integrating the logic network into microfluidic chips that provide rapid mixing and precise timing control. Third, real‑time monitoring of multilayer logic states will require high‑resolution electrochemical imaging (e.g., scanning electrochemical microscopy) or multiplexed amperometric arrays. Fourth, the binary output can be interfaced with digital electronics and machine‑learning algorithms to perform higher‑level decision making, error correction, and adaptive thresholding. Finally, the authors stress the importance of robust binary discrimination: establishing clear voltage or current cut‑offs, implementing noise‑filtering circuitry, and employing catalytic enzymes (HRP, GOx) that amplify the signal while preserving the logical integrity of the network.
In conclusion, by marrying enzymatic Boolean logic with advanced electrode technologies, the proposed digital biosensor paradigm transcends the analog, quantitative limitations of traditional biosensors. It offers a pathway to highly selective, low‑error, and context‑aware detection platforms suitable for clinical diagnostics, environmental monitoring, and food safety applications, positioning logic‑enabled biosensing as a cornerstone of next‑generation smart analytical devices.
Comments & Academic Discussion
Loading comments...
Leave a Comment