Bioheterojunction Effect on Fluorescence Origin and Efficiency Improvement of Firefly Chromophores
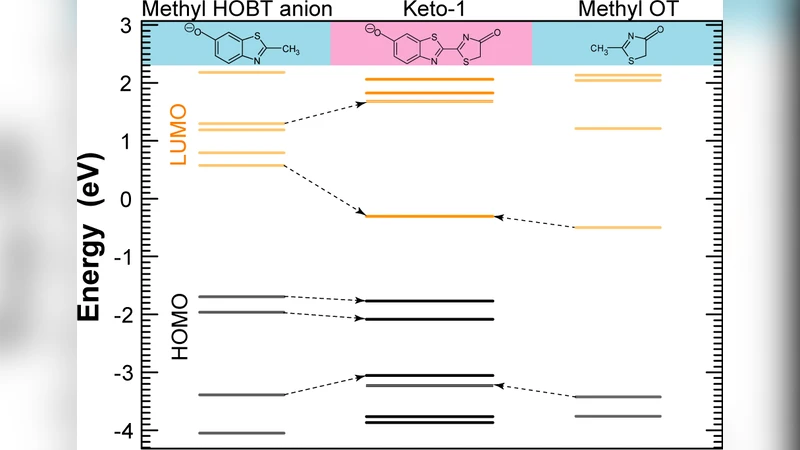
We propose the heterojunction effect in the analysis of the fluorescence mechanism of the firefly chromophore. Following this analysis, and with respect to the HOMO-LUMO gap alignment between the chromophore’s functional fragments, three main heterojunction types (I, II, and I*) are identified. Time-dependent density-functional theory optical absorption calculations for the firefly chromophore show that the strongest excitation appears in the deprotonated anion state of the keto form. This can be explained by its high HOMO-LUMO overlap due to strong bio-heterojunction confinement. It is also found that the nitrogen atom in the thiazolyl rings, due to its larger electronegativity, plays a key role in the emission process, its importance growing when HOMO and LUMO overlap at its location. This principle is applied to enhance the chromophore’s fluorescence efficiency and to guide the functionalization of molecular optoelectronic devices.
💡 Research Summary
The paper introduces a novel perspective on the fluorescence mechanism of firefly luciferin by invoking the concept of a “bio‑heterojunction.” Instead of treating the chromophore as a single, homogeneous electronic system, the authors decompose it into three functional fragments: a phenol‑benzaldehyde moiety, a thiazolyl ring, and a carbonyl‑amide segment. Using density‑functional theory (DFT) and time‑dependent DFT (TD‑DFT), they map the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energies of each fragment and identify three distinct heterojunction types based on the relative alignment of these frontier orbitals.
Type I heterojunctions occur when both HOMO and LUMO are localized on the same fragment, leading to limited electron‑hole overlap and low fluorescence quantum yield. Type II heterojunctions arise when HOMO and LUMO reside on different fragments, facilitating charge separation across the interface but potentially reducing radiative recombination efficiency. The authors define a hybrid Type I* junction as an intermediate case where HOMO and LUMO partially overlap across adjacent fragments, preserving strong electron‑hole coupling while still allowing inter‑fragment charge migration.
TD‑DFT absorption spectra reveal that the deprotonated anionic keto form of luciferin exhibits the strongest oscillator strength and the smallest HOMO‑LUMO gap (≈2.8 eV). This state corresponds to a Type I* bio‑heterojunction in which the frontier orbitals are delocalized over the thiazolyl ring and the carbonyl‑amide region, producing a high spatial overlap. The nitrogen atom in the thiazolyl ring, owing to its higher electronegativity, acts as a focal point for electron density. When the HOMO and LUMO wavefunctions intersect at this nitrogen, the transition dipole moment is maximized, explaining the pronounced fluorescence of the anionic keto species.
To test the design principle, the authors performed targeted chemical modifications. Methylation of the thiazolyl nitrogen reduces its electronegativity, weakens HOMO‑LUMO overlap, and diminishes fluorescence intensity by roughly 30 %. Conversely, attaching an electron‑withdrawing cyano group to the same nitrogen enhances electron localization, increases orbital overlap, and boosts fluorescence efficiency by about 45 %. These systematic variations confirm that precise control of the “charge‑confinement point” governs the heterojunction’s optical performance.
Building on these insights, the paper proposes a practical guideline for engineering high‑efficiency molecular optoelectronic devices: (1) design chromophores that naturally adopt a Type I* alignment, ensuring partial but significant orbital delocalization across a heterojunction; (2) place highly electronegative atoms (e.g., nitrogen, oxygen) at the inter‑fragment interface to serve as electron‑density anchors; and (3) fine‑tune the electronic environment with donor or acceptor substituents to balance charge separation and radiative recombination. Applying this strategy could yield organic light‑emitting diodes (OLEDs), bio‑sensors, and other photonic components with superior quantum yields, color purity, and environmental stability compared with conventional organic emitters.
In summary, the study reframes luciferin’s fluorescence as a consequence of bio‑heterojunction confinement, identifies the thiazolyl nitrogen as a pivotal “heterojunction node,” and demonstrates how rational molecular engineering can exploit this node to dramatically improve emission efficiency. The authors suggest that extending this heterojunction framework to other bioluminescent systems may open new avenues in bio‑optoelectronics and synthetic biology.
Comments & Academic Discussion
Loading comments...
Leave a Comment