Homeostatic competition drives tumor growth and metastasis nucleation
We propose a mechanism for tumor growth emphasizing the role of homeostatic regulation and tissue stability. We show that competition between surface and bulk effects leads to the existence of a critical size that must be overcome by metastases to reach macroscopic sizes. This property can qualitatively explain the observed size distributions of metastases, while size-independent growth rates cannot account for clinical and experimental data. In addition, it potentially explains the observed preferential growth of metastases on tissue surfaces and membranes such as the pleural and peritoneal layers, suggests a mechanism underlying the seed and soil hypothesis introduced by Stephen Paget in 1889 and yields realistic values for metastatic inefficiency. We propose a number of key experiments to test these concepts. The homeostatic pressure as introduced in this work could constitute a quantitative, experimentally accessible measure for the metastatic potential of early malignant growths.
💡 Research Summary
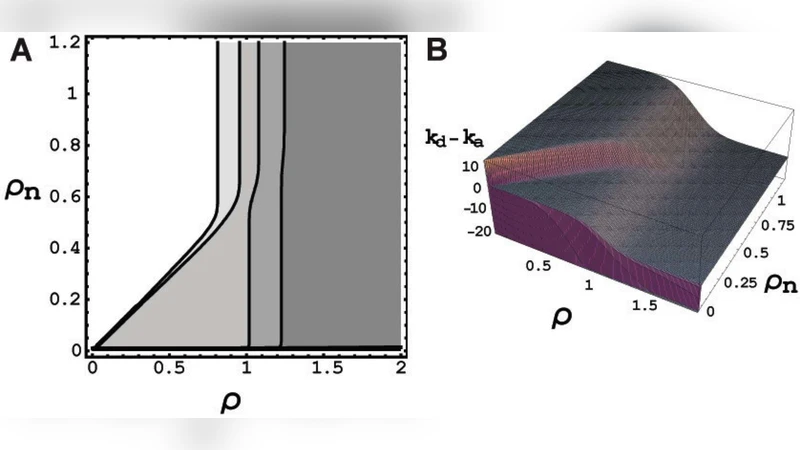
The paper introduces a physics‑based framework for tumor growth and metastasis that centers on the concept of homeostatic pressure—the intrinsic pressure a tissue exerts to maintain its volume and mechanical stability. By modeling a tumor as a body that experiences both a bulk‑driven proliferative pressure (driven by cell division) and a surface‑driven resistive tension (arising from tissue‑tumor interface mechanics), the authors derive a critical size (critical volume Vc) that a metastatic colony must exceed to continue expanding. Below this threshold, surface tension and the pressure differential between the tumor and surrounding normal tissue suppress net growth, leading to rapid regression or dormancy. Once Vc is surpassed, bulk proliferation dominates, and the lesion grows explosively.
Mathematically, the free‑energy of a tumor is expressed as F = (Ph – Pt)·V + γ·S, where Ph is the tumor‑generated internal pressure, Pt is the surrounding tissue pressure, γ is the interfacial surface tension, V is tumor volume, and S is its surface area. Minimization of F with respect to V and S yields the equilibrium condition and the critical volume Vc ≈ 2γ/(Ph – Pt). This simple relation links measurable mechanical parameters to the likelihood that a disseminated cell cluster will become a macroscopic metastasis.
The authors validate the model against clinical and experimental data showing that metastatic lesions are rarely observed below a certain size, and that the size distribution of detectable metastases follows a pattern consistent with a size‑dependent survival probability. Conventional models assuming a constant, size‑independent growth rate cannot reproduce these observations. Moreover, the framework naturally explains why metastases preferentially colonize thin, low‑tension surfaces such as pleura, peritoneum, and meninges: the reduced γ on these membranes lowers Vc, allowing even small clusters to establish growth.
Beyond descriptive power, the work proposes a quantitative biomarker—homeostatic pressure—that could be measured in vitro using three‑dimensional co‑culture systems combined with microfluidic pressure sensors. By embedding tumor cells within engineered normal tissue matrices and continuously monitoring pressure differentials, researchers can determine the pressure at which a nascent colony overcomes the critical threshold. This measurement would provide a direct estimate of metastatic potential for early lesions, offering a tool for risk stratification and personalized therapy planning.
The paper also reframes Stephen Paget’s “seed and soil” hypothesis in mechanical terms: the “seed” (cancer cell) carries an intrinsic proliferative pressure, while the “soil” (host tissue) supplies the mechanical landscape—its baseline pressure, elasticity, and interfacial tension—that determines whether the seed can germinate. Consequently, therapeutic strategies could target not only the cancer cells but also modify the mechanical properties of the host microenvironment. Potential interventions include agents that increase interfacial tension, drugs that stiffen the extracellular matrix, or physical therapies that alter tissue pressure gradients, thereby raising the critical size and reducing metastatic efficiency.
In summary, the study provides a coherent, experimentally testable model that integrates bulk proliferative forces and surface mechanical resistance to explain key features of tumor growth, metastasis nucleation, and organ‑specific colonization patterns. It offers a novel quantitative metric—homeostatic pressure—for assessing metastatic risk and suggests concrete experimental designs to validate and potentially exploit this metric in clinical oncology.
Comments & Academic Discussion
Loading comments...
Leave a Comment