Hysteresis in the cell response to time-dependent substrate stiffness
Mechanical cues like the rigidity of the substrate are main determinants for the decision making of adherent cells. Here we use a mechano-chemical model to predict the cellular response to varying substrate stiffness. The model equations combine the mechanics of contractile actin filament bundles with a model for the Rho-signaling pathway triggered by forces at cell-matrix contacts. A bifurcation analysis of cellular contractility as a function of substrate stiffness reveals a bistable response, thus defining a lower threshold of stiffness, below which cells are not able to build up contractile forces, and an upper threshold of stiffness, above which cells are always in a strongly contracted state. Using the full dynamical model, we predict that rate-dependent hysteresis will occur in the cellular traction forces when cells are exposed to substrates of time-dependent stiffness.
💡 Research Summary
In this study the authors develop a mechanochemical model that captures how adherent cells respond to changes in substrate stiffness, focusing on the emergence of bistability and rate‑dependent hysteresis in cellular contractility. The model couples two sub‑systems: (i) the mechanics of contractile actin‑myosin bundles, represented as elastic‑viscous elements that generate intracellular tension (σ) in response to substrate deformation (ε), and (ii) a biochemical signaling network centered on Rho‑GTPase activation. Mechanical forces at focal adhesions stimulate Rho, which in turn activates ROCK, leading to myosin light‑chain phosphorylation and enhanced actin‑myosin contractility, thus closing a positive feedback loop between force generation and signaling.
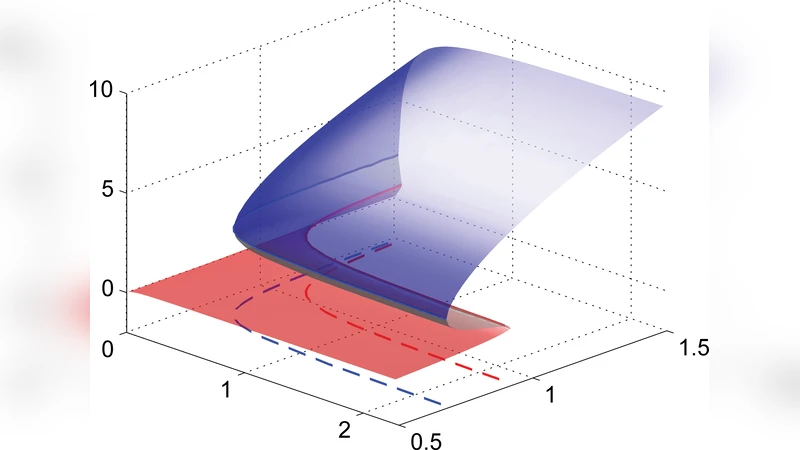
Mathematically the system is described by a set of nonlinear ordinary differential equations for σ, the Rho activity variable (R), and ε. A bifurcation analysis with substrate stiffness (k_sub) as the control parameter reveals two critical stiffness values, k_low and k_high. For k_sub < k_low the feedback loop cannot sustain tension, and the cell remains in a non‑contractile state (σ≈0). For k_sub > k_high the feedback is self‑reinforcing, driving the system to a high‑tension steady state. In the intermediate range k_low < k_sub < k_high both low‑tension and high‑tension equilibria coexist, giving rise to bistability; the final state depends on initial conditions or perturbations.
To explore dynamic environments the authors impose a time‑varying stiffness k_sub(t). Numerical simulations show that when stiffness is increased slowly, the transition to the high‑tension branch occurs at a stiffness lower than the static upper threshold, whereas a slow decrease triggers the return to the low‑tension branch at a stiffness higher than the static lower threshold. This path‑dependence creates a hysteresis loop in the traction force versus stiffness curve. The loop area grows with decreasing rate of stiffness change, indicating that the system’s intrinsic feedback delay can fully develop only under quasi‑static conditions; rapid stiffness variations suppress hysteresis.
The model’s predictions align with experimental observations of mechanical memory in stem cells, fibroblasts, and cancer cells, where cells retain a contractile phenotype after exposure to stiff substrates and require a substantial reduction in stiffness to revert. The bistable regime provides a mechanistic explanation for why cells can adopt distinct phenotypes under identical mechanical cues, while the hysteresis explains the observed lag in phenotype switching when the extracellular matrix remodels over time.
Overall, the paper offers a unified theoretical framework that integrates cytoskeletal mechanics with Rho‑mediated signaling, identifies lower and upper stiffness thresholds that delimit contractile behavior, and demonstrates that the rate of substrate stiffening or softening governs a hysteresis effect in cellular traction forces. These insights have practical implications for tissue engineering, regenerative medicine, and cancer metastasis, where designing substrates with controlled stiffness dynamics could steer cell fate and function.
Comments & Academic Discussion
Loading comments...
Leave a Comment