Phase Transition Induced Fission in Lipid Vesicles
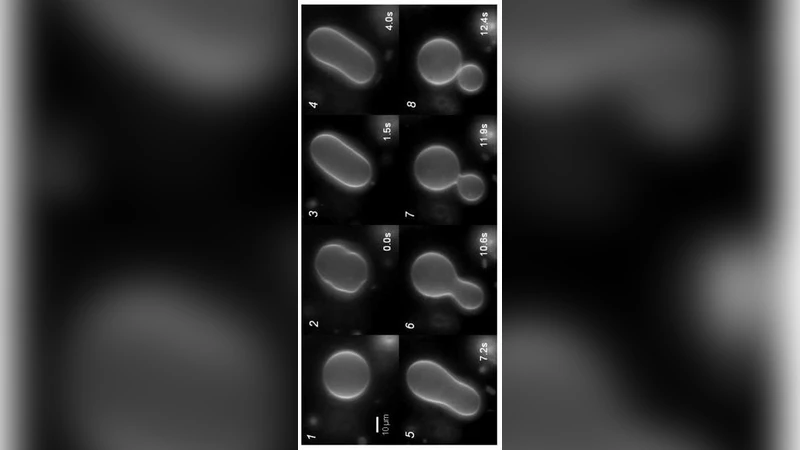
In this work we demonstrate how the first order phase transition in giant unilamellar vesicles (GUVs) can function as a trigger for membrane fission. When driven through their gel-fluid phase transition GUVs exhibit budding or pearl formation. These buds remain connected to the mother vesicle presumably by a small neck. Cooling these vesicles from the fluid phase (T>Tm) through the phase transition into the gel state (T<Tm), leads to complete rupture and fission of the neck, while the mother vesicle remains intact. Pearling tubes which formed upon heating break-up and decay into multiple individual vesicles which then diffuse freely. Finally we demonstrate that mimicking the intracellular bulk viscosity by increasing the bulk viscosity to 40cP does not affect the overall fission process, but leads to a significant decrease in size of the released vesicles.
💡 Research Summary
In this study the authors investigate how the first‑order gel‑fluid phase transition of giant unilamellar vesicles (GUVs) can be harnessed as a purely physical trigger for membrane fission. GUVs composed primarily of dipalmitoylphosphatidylcholine (DPPC), which has a well‑defined melting temperature (Tm ≈ 41 °C), were prepared with fluorescent markers to allow real‑time optical observation. When the vesicles were heated above Tm, the membrane entered a fluid state, expanded in area, and spontaneously formed buds and pearl‑like tubes. These protrusions remained attached to the parent vesicle by narrow necks whose radii were on the order of tens of nanometres; the necks are stabilized by a delicate balance between Laplace pressure and membrane bending elasticity.
Subsequent cooling through the transition back into the gel phase (T < Tm) caused a dramatic increase in membrane rigidity and shear resistance. The previously formed necks could no longer sustain the mechanical stress and ruptured, resulting in complete scission of the buds or tubes while the mother vesicle remained intact. The detached structures collapsed into individual vesicles that diffused freely in the surrounding medium. High‑speed imaging captured the rapid neck breakage and allowed quantitative measurement of the critical curvature, neck radius, and energy release associated with the fission event.
To test the influence of intracellular‑like bulk viscosity, the authors increased the viscosity of the external solution to ≈40 cP using glycerol‑based mixtures. This manipulation did not alter the fundamental fission pathway; however, it significantly reduced the average diameter of the released vesicles. The authors attribute this effect to the suppression of internal fluid flow within the budding tubes, which forces the neck to become thinner and shorter before rupture, thereby concentrating the released elastic energy into a smaller volume.
The work demonstrates that a temperature‑driven phase transition alone is sufficient to drive membrane scission without the need for protein machineries such as dynamin or external mechanical forces. By simply tuning the temperature ramp and the bulk viscosity, the size of the daughter vesicles can be finely controlled. This finding opens new avenues for the design of artificial cell division systems, targeted drug‑delivery carriers, and minimal protocell models. Moreover, the authors provide a quantitative framework linking membrane elastic parameters, curvature, and viscous dissipation to the observed fission threshold, offering a solid theoretical basis for future engineering of lipid‑based nanodevices.
Comments & Academic Discussion
Loading comments...
Leave a Comment