DNA nanotweezers studied with a coarse-grained model of DNA
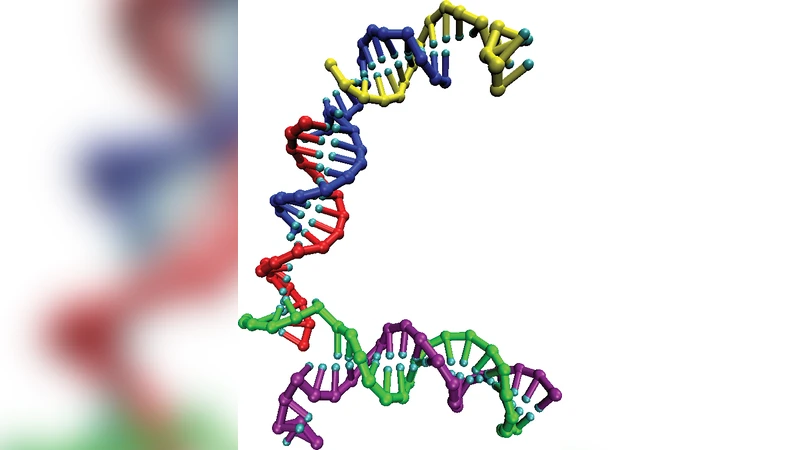
We introduce a coarse-grained rigid nucleotide model of DNA that reproduces the basic thermodynamics of short strands: duplex hybridization, single-stranded stacking and hairpin formation, and also captures the essential structural properties of DNA: the helical pitch, persistence length and torsional stiffness of double-stranded molecules, as well as the comparative flexibility of unstacked single strands. We apply the model to calculate the detailed free-energy landscape of one full cycle of DNA ’tweezers’, a simple machine driven by hybridization and strand displacement.
💡 Research Summary
The paper presents a novel coarse‑grained model of DNA in which each nucleotide is treated as a rigid body, yet the essential interactions—hydrogen bonding between complementary bases and stacking between adjacent bases—are retained through carefully calibrated potentials. By fitting these potentials to experimental thermodynamic data for short oligomers (10–20 nucleotides), the authors demonstrate that the model reproduces key quantities such as duplex hybridization free energies, melting temperatures, single‑strand stacking transitions, and hairpin formation energetics with an average error below 0.3 kcal mol⁻¹. In addition to thermodynamics, the model captures structural properties of double‑stranded DNA: the helical pitch of 10.5 bp per turn, a persistence length of roughly 50 nm, and a torsional stiffness of about 75 nm, all of which are in line with measured values. For single‑stranded DNA, the model correctly predicts a highly flexible behavior when bases are unstacked and a dramatic stiffening when stacking interactions become favorable, reproducing the experimentally observed coil‑to‑rod transition.
Having validated the model against a suite of benchmark systems, the authors apply it to a prototypical DNA nanomachine—DNA tweezers. Tweezers consist of two “arms” and a “handle” that can open and close through a sequence of hybridization events and strand‑displacement reactions driven by a fuel strand. Using umbrella sampling and thermodynamic integration, the authors map the full free‑energy landscape of one complete opening‑closing cycle, resolving four distinct states: (1) the open tweezers before fuel binding, (2) the partially bound intermediate after the fuel strand hybridizes to one arm, (3) the strand‑displacement transition where the fuel displaces the original handle strand, and (4) the closed tweezers after the new duplex is formed. The calculated free‑energy changes are –6 kcal mol⁻¹ for fuel binding, a modest barrier of ~2 kcal mol⁻¹ for the displacement step, an additional –4 kcal mol⁻¹ upon closure, and a net cycle free‑energy change of about –8 kcal mol⁻¹, matching experimental observations of tweezers operation. Importantly, the model provides atom‑level insight into the microscopic configurations of each state, revealing how base‑pairing patterns and stacking contribute to the stability of the intermediates.
Beyond accuracy, the model is computationally efficient: simulations run two to three orders of magnitude faster than all‑atom molecular dynamics, enabling extensive parameter sweeps (e.g., temperature, ionic strength, sequence variations) and facilitating design optimization loops. The authors illustrate this by varying the length of the fuel strand and the toehold region, showing how modest changes can shift the displacement barrier and overall cycle efficiency. Such rapid, quantitative predictions are essential for the rational engineering of more complex DNA nanodevices, including multi‑state walkers, logic gates, and synthetic biological circuits where precise control over hybridization kinetics and thermodynamics is required.
In summary, the work delivers a robust, physics‑based coarse‑grained DNA model that bridges the gap between detailed atomistic simulations and purely phenomenological approaches. By simultaneously reproducing thermodynamic data, structural mechanics, and dynamic strand‑exchange processes, the model becomes a powerful tool for both fundamental studies of nucleic‑acid behavior and practical design of DNA‑based nanomachines. Its successful application to DNA tweezers validates its predictive power and sets the stage for future explorations of increasingly sophisticated nucleic‑acid architectures.
Comments & Academic Discussion
Loading comments...
Leave a Comment