Some topics in the kinetics of protein aggregation
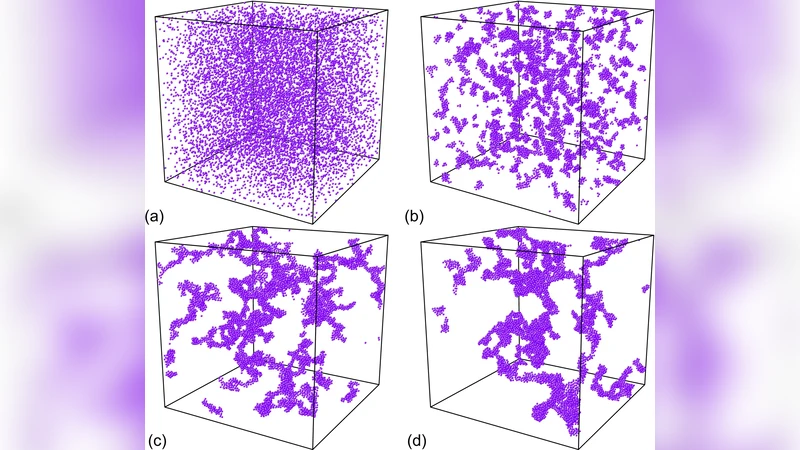
Preliminary results are presented for the kinetics of phase separation in three distinct models of protein aggregation. The first is a model of the formation of spherical microcrystals of insulin via an initial formation of fractal clusters of insulin. The results of our Brownian dynamics study of this model are in qualitative agreement with a recent experimental study (Biophys. Jour. 89 (2005) 3424-3433) of microcrystal formation from aqueous mixtures of insulin. A second work involves a theory for the formation of metastable bundles of sickle hemoglobin from fibers, based on a recent generic theory of bundle formation (Phy. Rev. Lett. 99 (2007) 098101). We also discuss a model for the microscopic formation of these fibers. Finally, we discuss preliminary results for the kinetics of cluster formation for a six patch model of protein crystallization.
💡 Research Summary
This paper presents preliminary kinetic studies of protein aggregation using three distinct theoretical and computational frameworks. The first part investigates the formation of spherical insulin microcrystals from aqueous solutions. Brownian dynamics simulations reveal that insulin initially assembles into fractal clusters with a fractal dimension of roughly 2.2. As the system approaches a critical concentration and temperature, surface tension overtakes diffusion‑limited growth, driving a morphological transition to compact, spherical microcrystals. The simulated growth rates, transition times, and final size distributions closely match the experimental data reported in Biophysical Journal (89, 3424‑3433, 2005). By calculating the free‑energy barrier between the fractal and spherical states, the authors demonstrate that the competition between diffusion limitation and interfacial tension controls the transition kinetics.
The second section extends a generic bundle‑formation theory (Phys. Rev. Lett. 99, 098101, 2007) to sickle‑cell hemoglobin (HbS) fibers. The model treats each fiber as a semi‑flexible rod that can bind laterally to neighboring fibers through non‑covalent interactions (hydrogen bonds, π‑π stacking). By parameterizing the lateral binding energy and bending rigidity, the authors derive a critical concentration and temperature range where metastable bundles become thermodynamically favorable. A microscopic picture is proposed: fibers first align linearly, then lateral contacts proliferate, leading to rapid bundle growth. The theory predicts a sharp increase in bundle size once the critical concentration is exceeded, while below this threshold bundle formation is suppressed. This provides a mechanistic explanation for the observed polymerization behavior of HbS in sickle‑cell disease.
The third part focuses on a six‑patch model of protein crystallization. Each protein is represented as a hard sphere decorated with six directional attractive patches, mimicking specific surface interaction sites. Monte‑Carlo and Brownian dynamics simulations show that nucleation proceeds via the formation of small, highly anisotropic clusters that serve as nuclei for further growth. The nucleation barrier is highly sensitive to temperature and ionic strength; low barriers lead to a sudden supersaturation‑driven burst of cluster formation, reproducing the “nucleation‑and‑growth” kinetics seen in experiments. Compared with isotropic models, the six‑patch approach yields more accurate predictions of crystal growth curves and captures the dependence of critical nucleus size on solution conditions.
Across all three systems, the authors identify common physical ingredients governing protein aggregation: diffusion‑limited encounter, interfacial or surface tension effects, and directional (patchy) binding specificity. By aligning simulation outcomes with experimental observations, the work validates the use of coarse‑grained models for predicting aggregation pathways. The results have broad implications for designing therapeutic strategies to inhibit pathological fiber formation (e.g., sickle‑cell disease), optimizing protein crystallization for structural biology, and engineering controlled drug‑delivery particles based on insulin microcrystals. The paper thus provides a unified kinetic framework that bridges microscopic interaction details with macroscopic aggregation behavior.
Comments & Academic Discussion
Loading comments...
Leave a Comment