History dependent dynamics in a generic model of ion channels - an analytic study
Recent experiments have demonstrated that the timescale of adaptation of single neurons and ion channel populations to stimuli slows down as the length of stimulation increases; in fact, no upper bound on temporal time-scales seems to exist in such systems. Furthermore, patch clamp experiments on single ion channels have hinted at the existence of large, mostly unobservable, inactivation state spaces within a single ion channel. This raises the question of the relation between this multitude of inactivation states and the observed behavior. In this work we propose a minimal model for ion channel dynamics which does not assume any specific structure of the inactivation state space. The model is simple enough to render an analytical study possible. This leads to a clear and concise explanation of the experimentally observed exponential history-dependent relaxation in sodium channels in a voltage clamp setting, and shows that their recovery rate from slow inactivation must be voltage dependent. Furthermore, we predict that history-dependent relaxation cannot be created by overly sparse spiking activity. While the model was created with ion channel populations in mind, its simplicity and genericalness render it a good starting point for modeling similar effects in other systems, and for scaling up to higher levels such as single neurons which are also known to exhibit multiple time scales.
💡 Research Summary
The paper addresses a striking experimental observation: both single neurons and populations of ion channels exhibit ever‑slowing adaptation when the duration of a stimulus is increased, suggesting that there is no intrinsic upper bound on the temporal scales involved. In particular, voltage‑clamp recordings of sodium channels reveal an exponential, history‑dependent relaxation that cannot be captured by the traditional two‑state (open/closed) or even a few‑state kinetic schemes. Moreover, single‑channel patch‑clamp studies hint at a vast, largely invisible space of inactivated states. To reconcile these findings, the authors propose a minimal, generic model that deliberately avoids any detailed assumption about the topology of the inactivation manifold.
The core of the model is a Markov chain with one conducting state and an infinite ladder of inactivated states. Each inactivated state is characterized by its own transition rate, and crucially these rates are functions of membrane voltage. By treating the ladder as a continuum, the authors replace the discrete set of states with a probability density over a continuous “depth” variable. The voltage dependence is introduced through a Boltzmann‑type factor: depolarization accelerates transitions from the conducting to deeper inactivated states while simultaneously slowing the reverse transitions.
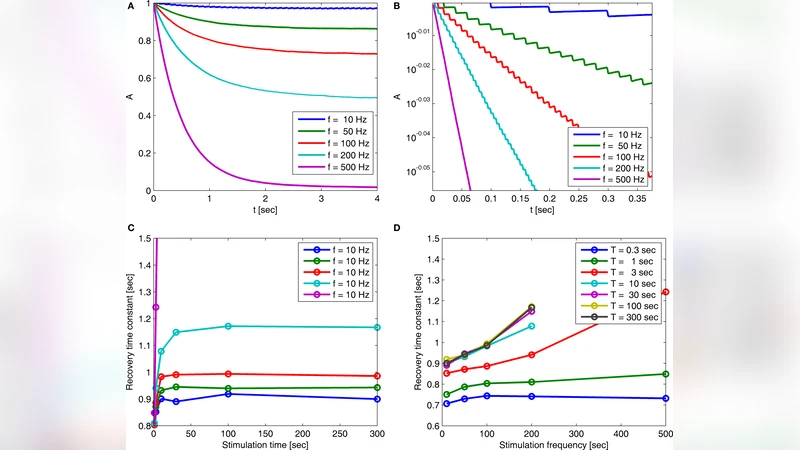
Using Laplace transforms, the authors derive a closed‑form expression for the system’s response function in the complex frequency domain. Pole analysis of this expression reveals a spectrum of exponential modes, each associated with a particular region of the inactivated‑state continuum. When the stimulus is brief, the fast poles dominate and the relaxation appears mono‑exponential. As stimulus duration grows, the contribution of the slowest pole—corresponding to the deepest inactivated states—becomes increasingly important, producing the experimentally observed long‑tail, history‑dependent decay. The analytical solution explicitly shows that the effective recovery rate is voltage‑dependent: at more hyper‑polarized potentials the deepest states are more readily exited, leading to faster recovery, whereas depolarized potentials trap channels in deep inactivation, slowing the return to the conducting state.
Beyond reproducing voltage‑clamp data, the model makes a novel prediction about the role of spiking activity. If spikes are sparse enough that the inter‑spike interval exceeds the characteristic time needed to populate deep inactivated states, the system never accumulates a significant population in those states, and the history‑dependent relaxation disappears. Conversely, high‑frequency spiking or sustained depolarization continuously drives channels into deeper inactivation, generating multi‑scale adaptation. This prediction aligns with observations that neurons subjected to irregular, low‑rate firing exhibit less pronounced adaptation than those driven by tonic high‑frequency input.
The authors validate the model by comparing its predictions with published sodium‑channel voltage‑clamp recordings. The fit captures both the shape of the recovery curve and its voltage dependence across a range of stimulus lengths, outperforming conventional multi‑state kinetic models that require ad‑hoc parameter tuning. Importantly, the model achieves this with far fewer free parameters because it does not need to specify the exact number or connectivity of inactivated states.
Finally, the paper discusses broader implications. Because the framework is agnostic to the microscopic details of the inactivation landscape, it can be readily extended to other ion‑channel families (e.g., calcium or potassium channels) and to higher‑level neuronal models where multiple time scales are observed (e.g., spike‑frequency adaptation, after‑hyperpolarization dynamics). The authors suggest that incorporating such a continuum‑inactivation component into single‑neuron or network models could provide a parsimonious explanation for the ubiquitous presence of slow processes in neural computation without resorting to phenomenological “hidden” variables.
In summary, the study offers a mathematically tractable, experimentally grounded model that links a vast, unobservable inactivation state space to the history‑dependent, voltage‑dependent relaxation observed in ion‑channel physiology. It clarifies why adaptation can span arbitrarily long time scales, predicts conditions under which such multi‑scale behavior is suppressed, and opens a pathway for integrating similar mechanisms into more comprehensive models of neuronal function.
Comments & Academic Discussion
Loading comments...
Leave a Comment