Consequences of cell-to-cell P-glycoprotein transfer on acquired multidrug resistance in breast cancer: a cell population dynamics model
Cancer is a proliferation disease affecting a genetically unstable cell population, in which molecular alterations can be somatically inherited by genetic, epigenetic or extragenetic transmission processes, leading to a cooperation of neoplastic cells within tumoral tissue. The efflux protein P-glycoprotein (P gp) is overexpressed in many cancer cells and has known capacity to confer multidrug resistance to cytotoxic therapies. Recently, cell-to-cell P-gp transfers have been shown. Herein, we combine experimental evidence and a mathematical model to examine the consequences of an intercellular P-gp trafficking in the extragenetic transfer of multidrug resistance from resistant to sensitive cell subpopulations. We report cell-to-cell transfers of functional P-gp in co-cultures of a P-gp overexpressing human breast cancer MCF-7 cell variant, selected for its resistance towards doxorubicin, with the parental sensitive cell line. We found that P-gp as well as efflux activity distribution are progressively reorganized over time in co-cultures analyzed by flow cytometry. A mathematical model based on a Boltzmann type integro-partial differential equation structured by a continuum variable corresponding to P-gp activity describes the cell populations in co-culture. The mathematical model elucidates the population elements in the experimental data, specifically, the initial proportions, the proliferative growth rates, and the transfer rates of P-gp in the sensitive and resistant subpopulations. We confirmed cell-to-cell transfer of functional P-gp. The transfer process depends on the gradient of P-gp expression in the donor-recipient cell interactions, as they evolve over time. Extragenetically acquired drug resistance is an additional aptitude of neoplastic cells which has implications in the diagnostic value of P-gp expression and in the design of chemotherapy regimens
💡 Research Summary
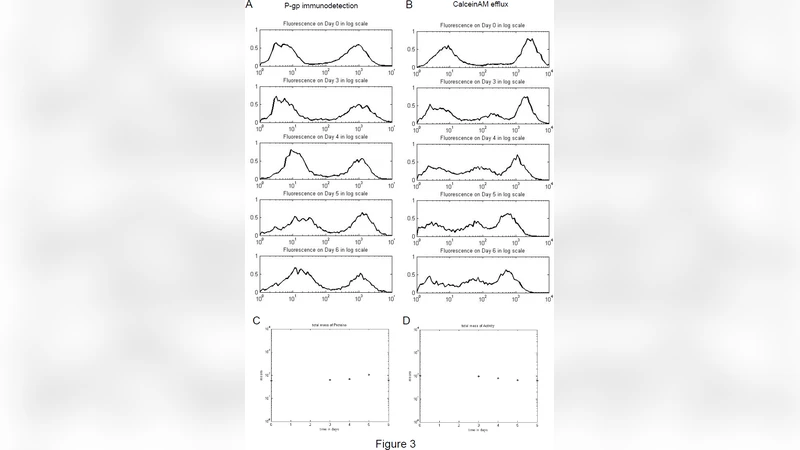
This study investigates how intercellular transfer of the drug‑efflux protein P‑glycoprotein (P‑gp) can spread multidrug resistance (MDR) within a breast‑cancer cell population. The authors used a doxorubicin‑resistant MCF‑7 variant that overexpresses functional P‑gp (designated MCF‑7/DOX) and co‑cultured it with the parental, drug‑sensitive MCF‑7 line at a 1:1 ratio. Over a period of up to 96 hours, they sampled the mixed cultures at several time points and measured surface P‑gp levels and efflux activity by flow cytometry. Initially, the two subpopulations were clearly distinct: the resistant cells displayed high fluorescence intensity for P‑gp antibodies and strong rhodamine‑123 efflux, whereas the sensitive cells showed low values. As co‑culture progressed, the sensitive subpopulation gradually acquired P‑gp, as evidenced by a right‑ward shift in fluorescence histograms and an increase in efflux capacity. By the end of the experiment, the overall distribution of P‑gp activity had become continuous, indicating that functional protein had moved from donor to recipient cells.
To interpret these observations quantitatively, the authors constructed a population‑level mathematical model based on a Boltzmann‑type integro‑partial differential equation (integro‑PDE). The state variable x represents a continuous measure of P‑gp activity (e.g., mean fluorescence intensity). The density n(t, x) denotes the number of cells at time t possessing activity level x. The model contains two principal terms: (i) a growth term g(x) n(t, x) that captures proliferation and death, allowing the net growth rate to depend on P‑gp expression; and (ii) a transfer term T
Comments & Academic Discussion
Loading comments...
Leave a Comment