A symmetry breaking mechanism for epithelial cell polarization
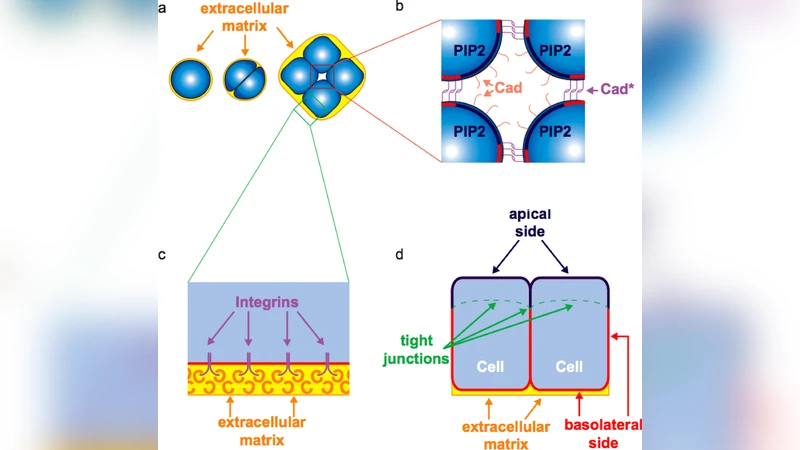
In multicellular organisms, epithelial cells form layers separating compartments responsible for different physiological functions. At the early stage of epithelial layer formation, each cell of an aggregate defines an inner and an outer side by breaking the symmetry of its initial state, in a process known as epithelial polarization. By integrating recent biochemical and biophysical data with stochastic simulations of the relevant reaction-diffusion system we provide evidence that epithelial cell polarization is a chemical phase separation process induced by a local bistability in the signaling network at the level of the cell membrane. The early symmetry breaking event triggering phase separation is induced by adhesion-dependent mechanical forces localized in the point of convergence of cell surfaces when a threshold number of confluent cells is reached. The generality of the emerging phase separation scenario is likely common to many processes of cell polarity formation.
💡 Research Summary
The paper investigates how epithelial cells break symmetry to establish polarity during the early stages of tissue formation. By assembling recent biochemical data on key membrane‑associated signaling components—PI3K, PTEN, PIP2, PIP3, and small GTPases such as Rac and Cdc42—the authors construct a reaction‑diffusion model that captures the essential feedback loops governing phosphoinositide conversion. The model predicts a bistable regime in which two distinct steady states coexist: a PIP3‑rich state and a PIP2‑rich state. Crucially, the transition from a homogeneous, symmetric configuration to a polarized one is triggered by a mechanical cue that arises when a sufficient number of cells become confluent. At points where multiple cell surfaces converge, adhesion‑mediated tension locally deforms the membrane, enhancing PI3K activity and thereby raising PIP3 concentration in a confined region. This local increase initiates a positive feedback loop that spreads, while PTEN‑mediated dephosphorylation sustains the complementary PIP2‑rich domain. The resulting spatial segregation mirrors a chemical phase‑separation process, with the membrane effectively demixing into two coexisting phases that define the outer (apical) and inner (basal) sides of each cell.
Stochastic simulations using Gillespie’s algorithm demonstrate that the polarity outcome is robust against intrinsic noise and variations in initial conditions, provided the mechanical stimulus exceeds a defined threshold. The authors link this threshold to the number of neighboring cells and the strength of cadherin‑based adhesion, offering a quantitative explanation for experimentally observed “polarization triggers” that occur only after a critical cell density is reached. The study further argues that this phase‑separation mechanism is not limited to epithelial polarity; similar bistable membrane dynamics coupled with mechanical inputs could underlie other polarity phenomena such as neuronal axon‑dendrite specification or immune cell front‑rear polarization.
Overall, the work presents a unified physical‑chemical framework in which epithelial cell polarity emerges from the interplay of membrane‑localized signaling bistability and adhesion‑generated mechanical forces. By framing polarity as a symmetry‑breaking phase transition, the authors provide testable predictions for future experiments and suggest new avenues for therapeutic manipulation of tissue organization, regeneration, and cancer metastasis.
Comments & Academic Discussion
Loading comments...
Leave a Comment