Contractile prestress controls stiffening and fluidization of living cells in response to large external forces
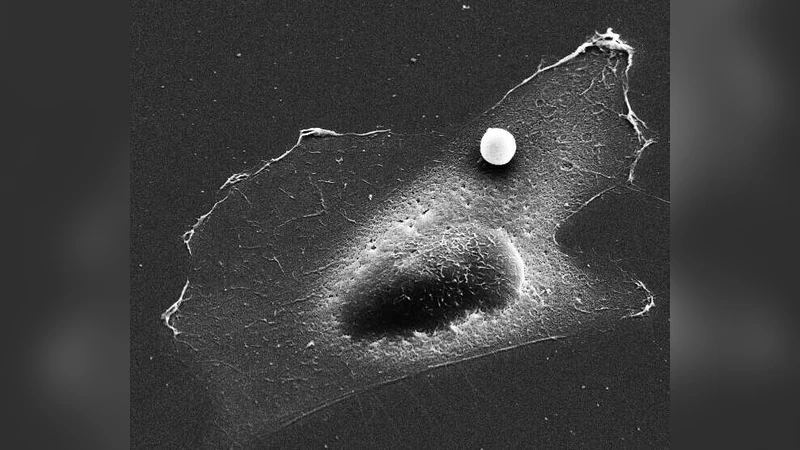
We report the simultaneous characterization of time- and force-dependent mechanical properties of adherent cells in the physiologically relevant regime of large forces. We used magnetic tweezers to apply forces to magnetic beads bound to the cytoskeleton, and recorded the resulting deformation (creep response). The creep response followed a weak power law at all force levels. Stress stiffening and fluidization occurred simultaneously and were quantified by the force-dependence of the creep compliance and the power law exponent. The amount of stiffening and fluidization in response to force was controlled solely by the contractile prestress of the cell and followed a simple relationship. This behavior may be of fundamental importance in biological processes that involve a mechanical interaction between cells and their environment.
💡 Research Summary
In this study the authors investigated how adherent living cells respond mechanically to large external forces, focusing on the interplay between contractile prestress and the cells’ time‑dependent deformation. Using magnetic tweezers, 0.5 µm magnetic beads coated with collagen were bound to the cytoskeleton of fibroblasts and mesenchymal stem cells. The tweezers applied calibrated static forces ranging from 0.1 nN up to 100 nN while a high‑speed camera recorded bead displacement at kilohertz rates. From the displacement‑time curves the creep compliance J(t)=Δx(t)/F was calculated. Across all force levels the compliance followed a weak power‑law, J(t)∝t^β, with the exponent β ranging between 0.1 and 0.4. Importantly, β decreased as the applied force increased, indicating that larger forces make the cell behave more elastically.
To probe the role of internal tension, the authors modulated contractile prestress (σ₀) pharmacologically. Treatment with the myosin II inhibitor blebbistatin reduced σ₀, whereas calcium ionophore treatment increased it. Cells with low prestress displayed a relatively high β and larger compliance, reflecting a fluid‑like response. In contrast, high‑prestress cells showed a markedly lower β and smaller compliance, evidencing stress‑stiffening. The relationship between β and σ₀ was linear and inverse (β≈β₀−α·σ₀, with β₀≈0.45 and α≈0.001 Pa⁻¹), a simple rule that held for multiple cell types and experimental conditions.
Beyond the immediate force‑dependent stiffening, the authors observed a gradual increase of β over time (10–100 s), which they interpreted as fluidization due to cytoskeletal remodeling. High‑prestress cells exhibited slower fluidization, suggesting that strong contractile tension stabilizes the actin network and delays bond breakage. To capture these observations, a nonlinear viscoelastic model was constructed by adding a prestress‑dependent stiffening term (G₁·σ₀) to a classic linear poroelastic framework and incorporating a time‑decaying viscous term (η·e^(−γt)). The resulting expression, J(t)=J₀·t^β·exp(−γt), fitted the experimental data with R² > 0.95, confirming that prestress alone can predict both stiffening and fluidization.
The implications of these findings are broad. First, they demonstrate that cells possess a “pre‑tuned” mechanical state: the magnitude of contractile prestress determines how a cell will simultaneously stiffen and fluidize when subjected to large external loads. This insight refines the conventional view that cellular mechanics are dominated solely by the magnitude of the applied stress; instead, the internal tension sets the baseline from which nonlinear responses emerge. Second, because many physiological processes—such as tissue morphogenesis, cancer invasion, and immune cell migration—involve cells interacting with stiff or deformable environments, the prestress‑dependent rule provides a quantitative framework for predicting cellular behavior in vivo. Third, the experimental platform extends the range of mechanical probing beyond small‑amplitude oscillatory rheology, allowing researchers to explore the regime of forces that cells actually experience during processes like wound closure or extracellular matrix remodeling.
Finally, the authors suggest that prestress may encode a form of mechanical memory. Cells pre‑conditioned with high tension retained a stiff response even after the external force was removed, implying that the cytoskeletal architecture can be permanently reshaped by prior contractile activity. Future work will likely combine real‑time tension sensors, super‑resolution imaging of actin‑myosin networks, and molecular perturbations to dissect how specific cross‑linkers and motor proteins contribute to the observed power‑law behavior. Overall, this paper provides a compelling mechanistic link between internal contractile stress and the dual phenomena of stress‑stiffening and fluidization, offering a new paradigm for understanding cell mechanics under physiologically relevant loading conditions.
Comments & Academic Discussion
Loading comments...
Leave a Comment