Extrinsic noise passing through a Michaelis-Menten reaction: A universal response of a genetic switch
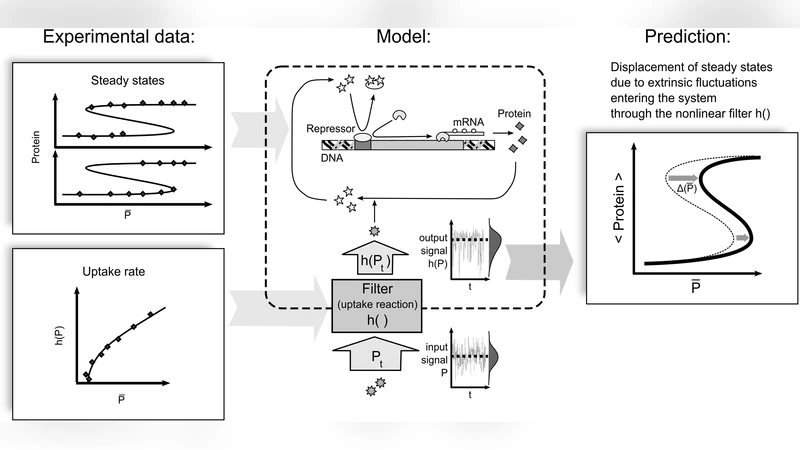
The study of biochemical pathways usually focuses on a small section of a protein interactions network. Two distinct sources contribute to the noise in such a system: intrinsic noise, inherent in the studied reactions, and extrinsic noise generated in other parts of the network or in the environment. We study the effect of extrinsic noise entering the system through a nonlinear uptake reaction which acts as a nonlinear filter. Varying input noise intensity varies the mean of the noise after the passage through the filter, which changes the stability properties of the system. The steady-state displacement due to small noise is independent on the kinetics of the system but it only depends on the nonlinearity of the input function. For monotonically increasing and concave input functions such as the Michaelis-Menten uptake rate, we give a simple argument based on the small-noise expansion, which enables qualitative predictions of the steady-state displacement only by inspection of experimental data: when weak and rapid noise enters the system through a Michaelis-Menten reaction, then the graph of the system’s steady states vs. the mean of the input signal always shifts to the right as noise intensity increases. We test the predictions on two models of lac operon, where TMG/lactose uptake is driven by a Michaelis-Menten enzymatic process. We show that as a consequence of the steady state displacement due to fluctuations in extracellular TMG/lactose concentration the lac switch responds in an asymmetric manner: as noise intensity increases, switching off lactose metabolism becomes easier and switching it on becomes more difficult.
💡 Research Summary
The paper investigates how extrinsic (environmental or network‑derived) noise influences the steady‑state behavior of biochemical systems when the noise enters through a nonlinear uptake reaction, specifically a Michaelis–Menten enzymatic step. The authors begin by distinguishing intrinsic noise—fluctuations generated by the stochastic nature of the reactions under study—from extrinsic noise, which originates elsewhere in the cell or in the surrounding environment. While intrinsic noise has been extensively modeled, the impact of extrinsic fluctuations on downstream modules is less well understood, especially when the signal passes through a nonlinear filter.
To address this gap, the authors employ a small‑noise (weak‑noise) expansion. Assuming the input variable (P) fluctuates rapidly around a mean (\langle P\rangle) with variance (\sigma^{2}) and that the uptake rate is a smooth function (h(P)), they expand the average output as (\langle h(P)\rangle \approx h(\langle P\rangle) + \frac{1}{2}h’’(\langle P\rangle)\sigma^{2}). For a Michaelis–Menten rate law (h(P)=V_{\max}P/(K_{m}+P)) the second derivative (h’’) is negative (the function is monotonically increasing and concave). Consequently, increasing the noise intensity reduces the effective uptake rate, shifting the system’s null‑cline to the right in the bifurcation diagram. Importantly, this shift depends only on the curvature of the input function and not on the specific kinetic parameters of the downstream network, making the effect universal for any system that receives its signal through such a filter.
The theoretical prediction is tested on two well‑known models of the lac operon. Both models incorporate TMG/lactose transport via a Michaelis–Menten transporter, but differ in the complexity of transcriptional regulation and feedback. Stochastic simulations are performed with varying noise amplitudes while keeping the mean extracellular TMG concentration fixed. The results confirm the analytical prediction: as the variance of the extracellular TMG concentration grows, the steady‑state curve of intracellular lac expression versus mean TMG shifts rightward. This shift produces an asymmetric hysteresis: the “off” transition (high TMG → low lac activity) occurs at lower TMG levels, whereas the “on” transition (low TMG → high lac activity) requires higher TMG concentrations. In other words, extrinsic fluctuations make it easier for the cell to shut down lactose metabolism and harder to reactivate it.
The authors emphasize that the effect hinges on the noise being fast compared with the intrinsic dynamics of the downstream module; slow fluctuations behave quasi‑statically and do not produce the same average shift. They also note that because the displacement is determined solely by the shape of the input function, experimentalists can predict the qualitative direction of the shift simply by inspecting the measured uptake curve, without detailed kinetic modeling.
In conclusion, the study reveals a universal mechanism by which extrinsic noise, filtered through a Michaelis–Menten reaction, alters the stability landscape of genetic switches. The displacement of steady states is independent of downstream kinetics and is dictated by the concave nature of the uptake function. This insight broadens our understanding of how cells integrate fluctuating environmental cues and suggests that many regulatory networks may exhibit similar noise‑induced bias whenever their inputs are processed by nonlinear enzymatic steps. Future work should explore other nonlinear filters (e.g., Hill functions) and quantify the role of noise spectra in shaping cellular decision‑making.