Ex-vivo detection of neural events using THz BioMEMS
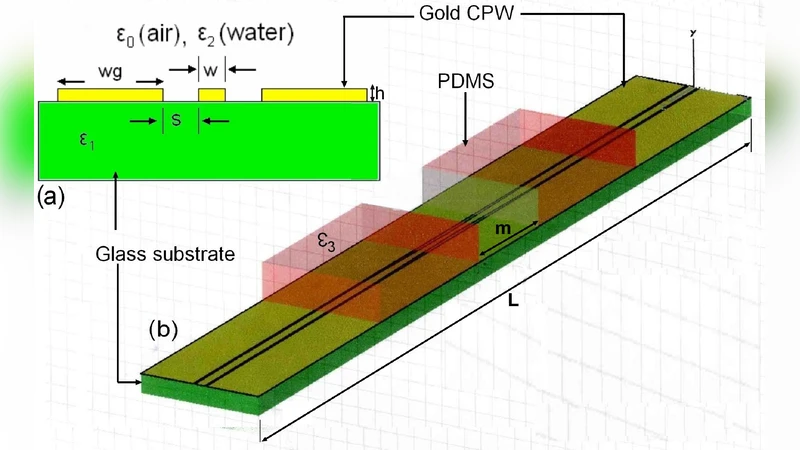
Background: Electromagnetic frequencies up to a few terahertz (THz) can yield real-time and noninvasive measurements on biological matter. Unfortunately, strong absorption in aqueous solutions and low spatial resolution return difficult free-space investigations. A new approach based on integrated THz circuits was used. The authors designed and fabricated a BioMEMS (Biological MicroElectro-Mechanical System) compatible with microfluidic circulation and electromagnetic propagation. It is dedicated to the ex vivo detection of nitric oxide synthase (NOS) activity, which is involved in neurodegenerative phenomena. Material/Methods: The biological model was a leech’s central nervous system. After its injury, the production of NO was observed and measured in the far-THz spectral domain. The nerve cord was put inside a BioMEMS realized in polydimethylsiloxane (PDMS) sealed on a glass wafer. Glass is a good material for supporting high-frequency integrated waveguides such as coplanar waveguides (CPWs). Measurements were performed with vectorial network analyser (VNA). Results: The transmission parameter in the frequency range of 0.14-0.22 THz was measured through CPWs located just below the microchannel containing the injured leech nerve cord. The lesion caused a decreased transmission coefficient due to the NOS activity. L-NAME was injected inside the microchannel and it was verified that it inhibits this activity. Conclusions: It was demonstrated that THz spectroscopy can detect a biochemical event, such as NOS activity around an injured leech’s nerve cord, in real time. Future studies will be dedicated to quantitative measurements of the reaction products using the sophisticated management of several drugs allowed with microfluidic microsystems.
💡 Research Summary
The paper presents a novel approach for real‑time, non‑invasive detection of biochemical events in biological tissue using terahertz (THz) spectroscopy integrated with a microfluidic BioMEMS platform. Conventional free‑space THz spectroscopy suffers from strong absorption in aqueous media, limiting its applicability to wet biological samples such as nervous tissue. To overcome this limitation, the authors designed a hybrid device that combines a coplanar waveguide (CPW) fabricated on a low‑loss glass substrate with a polydimethylsiloxane (PDMS) microchannel sealed on top. The glass substrate supports high‑frequency propagation (0.14–0.22 THz) with minimal insertion loss, while the PDMS channel provides a biocompatible environment for fluid handling, tissue placement, and precise drug delivery.
The biological model employed is the central nervous system (CNS) of the leech, chosen for its relatively large nerve cord that can be easily positioned inside the microchannel. After a controlled mechanical lesion, nitric oxide synthase (NOS) becomes active, producing nitric oxide (NO) and related reaction products that alter the dielectric properties of the surrounding medium. These changes affect the amplitude of the THz signal transmitted through the CPW beneath the channel. Using a vector network analyzer (VNA), the authors recorded the S21 transmission parameter before injury, immediately after injury, and after pharmacological intervention. The injury caused a measurable decrease in transmission (approximately 3–5 dB), indicating increased absorption or scattering due to NOS activity. Subsequent injection of the NOS inhibitor L‑NAME restored the transmission level to near‑baseline, confirming that the observed THz attenuation is directly linked to enzymatic activity.
The study demonstrates two key technical advances. First, the integration of a microfluidic PDMS layer with a glass‑based CPW enables THz interrogation of highly aqueous samples without the severe loss typical of free‑space measurements. Second, the system can monitor a specific enzymatic reaction (NOS activation) in real time, providing a label‑free, non‑destructive readout of a neurodegenerative‑related biochemical process. This capability opens new avenues for neurobiology research, drug screening, and potentially clinical diagnostics where rapid assessment of tissue health is required.
Future work outlined by the authors includes expanding the frequency range to capture richer spectral information, implementing multi‑channel designs for parallel monitoring of several tissue sites, and developing quantitative models that correlate THz transmission changes with absolute concentrations of NO and other reaction products. Moreover, adapting the platform to more complex animal models or human tissue samples will require optimization of channel geometry, CPW dimensions, and material choices to maintain low loss while ensuring biocompatibility. Ultimately, the authors envision that THz‑based BioMEMS could become a versatile platform for high‑resolution, real‑time biochemical sensing in a variety of biomedical applications.
Comments & Academic Discussion
Loading comments...
Leave a Comment