Chemical and Mechanical Signaling in Epithelial Spreading
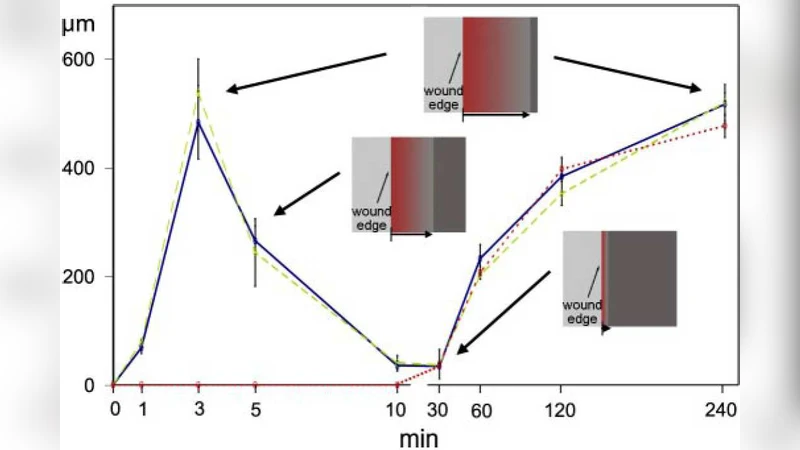
We propose a minimal mathematical model to explain long-range coordination of dynamics of multiple cells in epithelial spreading, which may be induced, under different conditions, by a chemical signal, or mechanical stress, or both. The model is based on chemo-mechanical interactions including a chemical effect of stress and concentration-dependent traction. The results, showing kinase concentration distribution and cell displacement, allow us to reproduce two activation waves on different time scales observed in the experiment, and distinguish between distinct dynamical patterns observed under conditions of injury or unconstraining.
💡 Research Summary
The paper addresses the long‑range coordination observed when epithelial sheets spread after injury or when mechanical constraints are released. Existing explanations have largely treated chemical cues (e.g., growth‑factor diffusion) and mechanical cues (e.g., tissue tension) in isolation, yet experiments reveal that both signals act together, producing two distinct activation waves: a fast, injury‑triggered wave and a slower, sustained wave that propagates across the monolayer. To capture this dual‑wave behavior, the authors construct a minimal chemo‑mechanical model that couples a continuum mechanics description of tissue stress with a reaction‑diffusion equation for a key intracellular kinase (or “signal” molecule).
Key elements of the model:
-
Mechanical field (σ) – The epithelial sheet is treated as a thin elastic layer. The local traction force exerted on the substrate, T, depends on the intracellular kinase concentration c. The balance of forces yields a partial differential equation for the stress σ(x,t), which diffuses through the tissue and is influenced by boundary conditions (fixed for “injury”, free for “unconstraining”).
-
Chemical field (c) – The concentration of an activated kinase obeys a reaction‑diffusion equation: ∂c/∂t = D∇²c + k₁σc – k₂c. The term k₁σc represents the stress‑induced activation (the “chemical effect of stress”), i.e., mechanical tension enhances the biochemical activation rate.
-
Traction‑concentration coupling – The traction generated by a cell is not constant; it rises with c but saturates, modeled as T(c)=T₀·c/(c+K). This captures the experimentally observed concentration‑dependent contractility and introduces a nonlinear feedback loop: higher c → larger traction → higher σ → further c activation.
The authors solve the coupled equations numerically in one‑ and two‑dimensional domains, initializing the system with a localized stress spike (mimicking injury) and a modest increase in c. The simulations generate two waves that match experimental observations:
-
Fast wave – Triggered almost instantaneously by the stress spike, it travels at ≈10 µm min⁻¹, consistent with the rapid calcium or MAPK wave seen in wound‑healing assays. This wave is primarily mechanical; its amplitude and speed are controlled by the stress‑activation coefficient (α) and the elastic modulus of the sheet.
-
Slow wave – After the fast wave decays, the diffusion of activated kinase together with the feedback from traction produces a slower front (≈1 µm min⁻¹) that sweeps the entire monolayer. This wave reflects the chemical propagation component and persists as long as the traction‑induced stress remains above the activation threshold.
By altering boundary conditions, the model reproduces distinct patterns observed under “injury” (fixed edges) versus “unconstraining” (free edges). In the injury case, the fast wave is damped because the fixed boundary suppresses stress buildup, and the slow wave dominates the dynamics. In the unconstrained case, stress can build up freely, leading to a pronounced fast wave that propagates simultaneously across the sheet, followed by the slower wave.
Parameter sensitivity analysis shows that the stress‑to‑chemical coupling coefficient (α) and the saturation constant K in the traction law are the most influential. Large α amplifies the fast wave, while a small K causes rapid saturation of traction, limiting the slow wave and slowing overall tissue remodeling. These findings suggest that epithelial cells may employ intrinsic feedback mechanisms to prevent runaway contractility while still enabling rapid coordination after injury.
The study’s contributions are threefold:
-
Unified framework – It provides a compact set of equations that simultaneously capture mechanical and chemical signaling, offering a quantitative bridge between disparate experimental observations.
-
Mechanistic insight – The model clarifies how mechanical stress can act as a catalyst for biochemical activation and how the resulting biochemical state feeds back to modulate tissue mechanics, producing the observed dual‑wave dynamics.
-
Predictive utility – By adjusting parameters, the model can predict how changes in substrate stiffness, cell contractility, or inhibitor treatments would alter wave speed and pattern, guiding future experimental designs in wound healing, tissue engineering, and cancer invasion studies.
Future extensions proposed by the authors include incorporating three‑dimensional geometry, nonlinear elastic responses, and cell‑cell adhesion dynamics, as well as coupling the model to live‑imaging data for real‑time parameter inference. Overall, the paper delivers a concise yet powerful theoretical tool for dissecting the intertwined chemical‑mechanical choreography that underlies epithelial spreading.
Comments & Academic Discussion
Loading comments...
Leave a Comment