Memristive model of amoebas learning
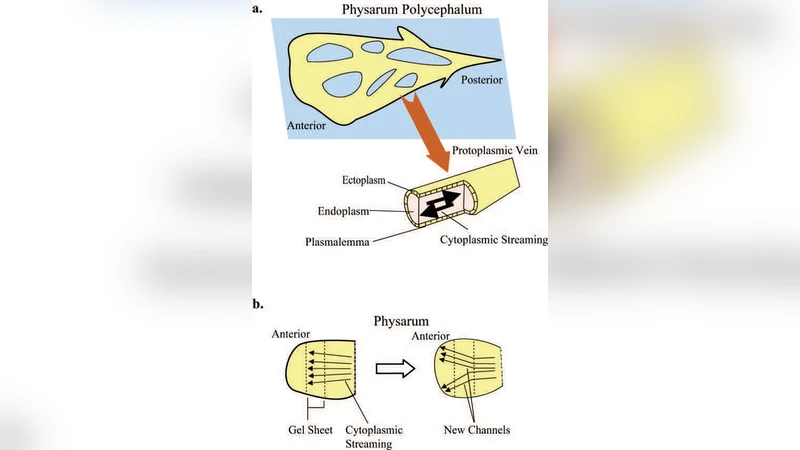
Recently, it was shown that the amoeba-like cell {\it Physarum polycephalum} when exposed to a pattern of periodic environmental changes learns and adapts its behavior in anticipation of the next stimulus to come. Here we show that such behavior can be mapped into the response of a simple electronic circuit consisting of an $LC$ contour and a memory-resistor (a memristor) to a train of voltage pulses that mimic environment changes. We also identify a possible biological origin of the memristive behavior in the cell. These biological memory features are likely to occur in other unicellular as well as multicellular organisms, albeit in different forms. Therefore, the above memristive circuit model, which has learning properties, is useful to better understand the origins of primitive intelligence.
💡 Research Summary
The paper addresses the intriguing observation that the slime‑mold Physarum polycephalum can “learn” periodic environmental changes and anticipate future stimuli, despite lacking a nervous system. The authors propose that this behavior can be captured by a very simple electronic analog: a series connection of an LC resonant circuit and a memory‑resistor (memristor). In this model, the LC part provides an oscillatory response to each voltage pulse that mimics an environmental perturbation, while the memristor supplies a non‑volatile, history‑dependent resistance that encodes the timing of the pulses.
When a train of voltage pulses with a fixed period is applied, the memristor’s resistance gradually decreases because the current flowing through it modifies its internal state variable (often modeled as the width of a doped region or the concentration of mobile ions). As the resistance drops, the quality factor (Q) of the LC circuit rises, allowing the circuit to sustain free oscillations at the same period even after the external pulses cease. This persistent oscillation reproduces the slime‑mold’s “anticipatory” contraction‑expansion cycles observed experimentally.
The authors link the memristive behavior to plausible biological mechanisms. They argue that the cell membrane’s ion channels and the cytoskeletal actin‑myosin network together generate a nonlinear, history‑dependent conductance. Periodic stimuli alter the membrane potential, driving Ca²⁺ influx and subsequent biochemical cascades that remodel the cytoskeleton. These structural changes affect both electrical conductivity and mechanical stiffness, creating a biophysical “state” that relaxes slowly—exactly the kind of dynamics captured by a memristor’s state variable.
To substantiate the analogy, the paper presents SPICE simulations of the LC‑memristor circuit. By varying pulse amplitude and period, the simulated memristor resistance curves match the experimentally measured adaptation curves of Physarum. The simulations demonstrate two key phenomena: (1) after three to four repetitions of a periodic pulse, the memristor’s resistance drops sharply, and (2) once the pulse train stops, the circuit voltage continues to oscillate at the learned period. These results quantitatively mirror the organism’s ability to predict the next stimulus.
The study yields two major insights. First, it suggests that primitive learning in unicellular organisms can be described by the same mathematical formalism used for modern memristive devices, implying that ion‑based memory is a universal principle across biology and electronics. Second, it shows that a single memristor coupled to a resonant element can store temporal patterns without any neural architecture, offering a blueprint for ultra‑low‑power artificial intelligence hardware that mimics biological anticipation.
Future directions outlined by the authors include (i) experimental integration of actual memristive materials (e.g., transition‑metal oxides, ion‑conducting polymers) with living cells to create hybrid bio‑electronic learning platforms, (ii) scaling the model to networks of memristors to emulate more complex adaptive behaviors such as multi‑frequency learning or conditional responses, and (iii) investigating whether similar ion‑based memory mechanisms exist in other unicellular and multicellular organisms, thereby probing the evolutionary roots of primitive intelligence. In sum, the LC‑memristor circuit provides a compelling, physically grounded framework for interpreting and reproducing the learning‑like dynamics observed in Physarum polycephalum.
Comments & Academic Discussion
Loading comments...
Leave a Comment