New dynamics in cerebellar Purkinje cells: torus canards
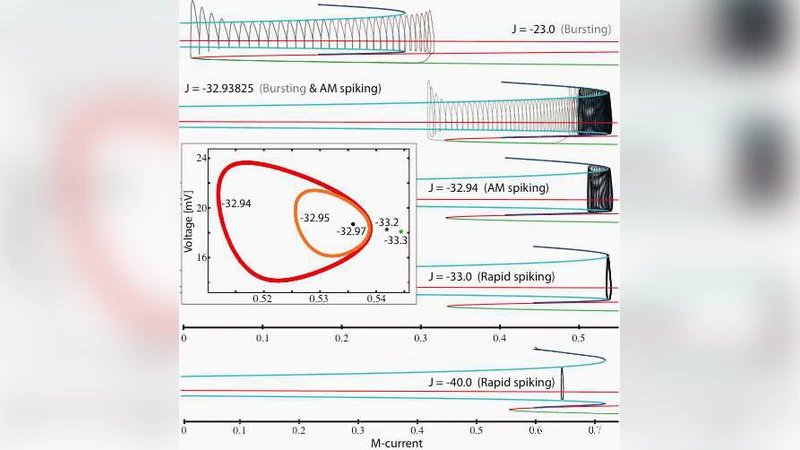
We describe a transition from bursting to rapid spiking in a reduced mathematical model of a cerebellar Purkinje cell. We perform a slow-fast analysis of the system and find that – after a saddle node bifurcation of limit cycles – the full model dynamics follow temporarily a repelling branch of limit cycles. We propose that the system exhibits a dynamical phenomenon new to realistic, biophysical applications: torus canards.
💡 Research Summary
The paper investigates a novel dynamical phenomenon that occurs during the transition from bursting to rapid spiking in cerebellar Purkinje cells. Starting from a detailed biophysical model comprising 559 compartments and over 6,000 state variables, the authors reduce the system to a five‑dimensional set of ordinary differential equations that capture the essential ionic currents: a delayed‑rectifier potassium current, a transient sodium current, a leak current, a high‑threshold calcium current, and a muscarinic‑suppressed potassium (M‑) current. The M‑current evolves on a timescale at least ten times slower than the other gating variables and therefore serves as the slow variable in a classic slow‑fast decomposition.
By varying an injected current parameter J, the authors map out the system’s behavior using continuation software (AUTO, XPPAUT). For relatively depolarizing values (J > ‑22.5 mV) the model settles to a stable fixed point. As J is decreased, bursts of rapid spikes appear, separated by long quiescent intervals. Near a critical value J* ≈ ‑32.938 mV the dynamics change qualitatively: the cell exhibits bursts interspersed with amplitude‑modulated (AM) fast spiking. During each AM cycle the slow M‑current slowly climbs along the branch of attracting limit cycles of the fast subsystem, reaches a fold of limit cycles, and then the full system follows temporarily the repelling branch of limit cycles before returning either to the attracting fixed‑point branch (ending a burst) or to the attracting limit‑cycle branch (producing another AM cycle). The number of AM cycles between bursts is highly sensitive to J and can vary from zero to dozens, reflecting a delicate balance between the slow M‑current and the fast calcium‑driven depolarization.
Further decreasing J eliminates the bursting altogether, leaving only AM‑modulated spiking, and eventually pure rapid spiking when J < ‑32.96 mV. The transition from AM spiking to unmodulated spiking coincides with a supercritical torus bifurcation: a stable invariant torus and an unstable limit cycle collide, giving rise to a stable periodic orbit whose Floquet multipliers cross the unit circle as J passes through the bifurcation value. This torus bifurcation provides the scaffold for the observed “torus canard” behavior.
The term “torus canard” is introduced to describe canard‑like trajectories that follow repelling branches of limit cycles rather than repelling fixed points, as in classical two‑dimensional canard theory (e.g., van der Pol). In the present five‑dimensional system, the canard is initiated not at a Hopf bifurcation but after a torus bifurcation, and it traverses the fold of limit cycles in the fast subsystem. Consequently, the system spends a finite amount of time near the unstable limit‑cycle branch, with the duration controlled by the parameter J, before returning to a stable manifold. This mechanism extends the traditional canard concept to higher‑dimensional, physiologically realistic neuronal models.
Physiologically, the M‑current hyperpolarizes the cell and opposes spiking, while the calcium current depolarizes and promotes spiking. When J > J*, the hyperpolarizing influence dominates, leading to burst termination and a quiescent phase. When J < J*, the depolarizing calcium drive overwhelms the M‑current, suppressing the quiescent phase and yielding continuous AM‑modulated spiking or pure spiking as the M‑current weakens further. Blocking either the M‑current or the calcium current in the reduced model reproduces the loss of AM modulation, a result confirmed in the full detailed model.
The authors suggest experimental verification by manipulating the M‑current pharmacologically (e.g., with muscarinic antagonists) or by current‑clamp protocols that mimic changes in J. Observations of slow amplitude modulation of fast spikes in vitro (as reported in previous Purkinje cell recordings) provide indirect support for the torus canard hypothesis.
In summary, the paper identifies and characterizes torus canards—canard trajectories that follow repelling limit‑cycle branches—in a realistic Purkinje cell model. By combining slow‑fast analysis, bifurcation continuation, and numerical simulations, the study elucidates how the interplay between a slow M‑current and a fast calcium current can generate complex mixed‑mode oscillations, including bursting, amplitude‑modulated spiking, and pure rapid spiking. The work expands the theoretical toolbox for neuronal dynamics, highlighting that canard phenomena are not confined to low‑dimensional systems with fixed‑point manifolds but can also arise in high‑dimensional biophysical models through torus bifurcations. Future directions include more sophisticated dimensional reduction techniques, rigorous analysis of the associated Poincaré maps, and exploration of chaotic regimes that may accompany the torus canard transition.
Comments & Academic Discussion
Loading comments...
Leave a Comment