Melting and freezing of argon in a granular packing of linear mesopore arrays
Freezing and melting of Ar condensed in a granular packing of template-grown arrays of linear mesopores (SBA-15, mean pore diameter 8 nanometer) has been studied by specific heat measurements C as a function of fractional filling of the pores. While interfacial melting leads to a single melting peak in C, homogeneous and heterogeneous freezing along with a delayering transition for partial fillings of the pores result in a complex freezing mechanism explainable only by a consideration of regular adsorption sites (in the cylindrical mesopores) and irregular adsorption sites (in niches of the rough external surfaces of the grains, and at points of mutual contact of the powder grains). The tensile pressure release upon reaching bulk liquid/vapor coexistence quantitatively accounts for an upward shift of the melting/freeezing temperature observed while overfilling the mesopores.
💡 Research Summary
This paper presents a detailed calorimetric investigation of the melting and freezing behavior of argon confined within the linear mesopores of SBA‑15 silica. SBA‑15 is a highly ordered mesoporous material consisting of hexagonally packed cylindrical pores with a mean diameter of 8 nm (pore radius R₀≈3.8 nm) and a narrow size distribution. The authors prepared a granular powder of SBA‑15 grains (≈1 µm) that possess a rough external surface, providing both regular adsorption sites (the interior of the cylindrical pores) and irregular sites (niches on the grain surface and contact points between grains).
Using a custom low‑rate (0.05 K min⁻¹) specific‑heat apparatus, the team measured the heat capacity C as a function of temperature for twelve different fractional fillings f (where f = n/n₀, n₀ is the amount required for complete pore filling). The samples were first saturated at 86 K, then cooled to 50 K (deep in the solid phase) and reheated to 86 K. For f ≥ 0.45 distinct anomalies in C were observed on both cooling and heating; for f ≤ 0.38 the signal was essentially absent, indicating that only the capillary‑condensed fraction participates in collective phase transitions.
Melting appears as a single, asymmetric peak centered at ≈74.5 K for f < 1, with a width of about 2 K. This behavior is interpreted in terms of interfacial (or surface) melting: a solid–liquid interface nucleates at the pore wall and propagates radially inward, producing a characteristic asymmetric heat‑capacity signature. The narrow pore‑size distribution of SBA‑15 eliminates the need to invoke a broad distribution of melting temperatures.
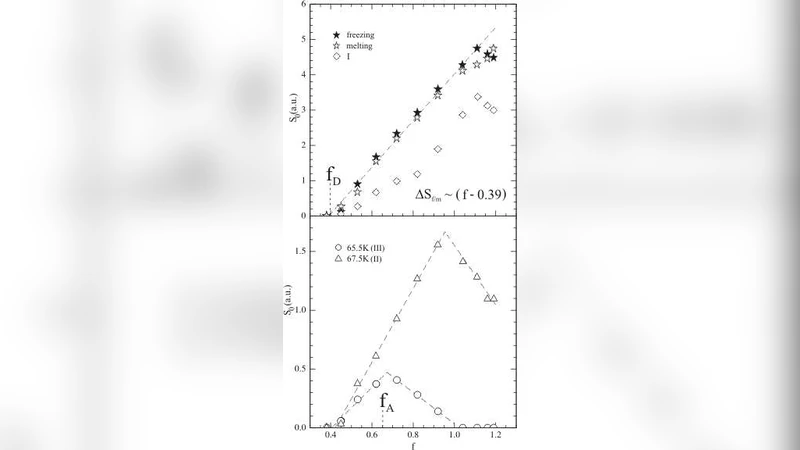
Freezing, by contrast, exhibits a complex, three‑component structure (labeled I, II, III). Component III, observed only for f < 1, corresponds to a “delayering” transition: the thin liquid film that initially coats the pore walls solidifies before the bulk capillary condensate. Component II is a sharp peak arising from homogeneous nucleation within the regular pores; its narrowness reflects the tight pore‑size distribution and its occurrence near the theoretical spinodal temperature T₋ (the lower limit of metastable liquid). Component I is a broad feature attributed to heterogeneous nucleation at irregular sites (surface roughness, grain contacts, and external niches). The relative weight of these components varies with filling fraction: at low f the heterogeneous contribution dominates, while near f ≈ 1 the homogeneous peak becomes prominent, and at still higher f the delayering contribution fades.
The entropy of fusion S₀, obtained by integrating the excess heat capacity over temperature, scales linearly with f for f < 1, extrapolating to zero at f ≈ 0.39, close to the desorption point of the sorption isotherm. This indicates that the “mobile” fraction—comprising both capillary‑condensed liquid and the wall‑coating film—freezes and melts collectively. Decomposing S₀ into the three freezing components reproduces the observed f‑dependence: component III follows the pore‑filled volume, component II reflects homogeneous nucleation, and component I grows with the probability of encountering heterogeneous nuclei.
When the pores are overfilled (f > 1), the liquid inside experiences a tensile (negative) pressure of about –70 ± 10 bar, as dictated by the Laplace pressure of concave menisci. Upon overfilling, the curvature of the meniscus disappears, releasing this tensile stress. Based on known pressure dependence of argon’s melting line, the authors predict—and experimentally confirm—a melting‑temperature increase of ≈1.5 K. A similar upward shift is observed for the onset of freezing in the overfilled regime, supporting the tensile‑pressure hypothesis. The paper also notes that water, with an anomalous melting‑pressure curve, shows a downward shift under analogous conditions, further validating the model.
Partial cooling–heating cycles were performed at f = 0.66. In one protocol, cooling was stopped at temperatures within the freezing range, solidifying only part of the liquid; subsequent heating reproduced the same melting peak shape but with reduced area, proportional to the amount solidified. In a second protocol, the sample was first fully solidified, then partially melted, and finally recooled. This sequence enhanced the freezing onset signal because the remaining solid acted as a nucleation seed, eliminating the need for heterogeneous nucleation. Notably, component III (delayering) reappeared in all partial cycles, whereas component II vanished when the isolated liquid parcels responsible for homogeneous nucleation were not regenerated.
The authors conclude that the freezing behavior in granular SBA‑15 is governed by a delicate interplay of homogeneous nucleation, heterogeneous nucleation, delayering transitions, and mechanical coupling via tensile pressure. Melting, on the other hand, is dominated by interfacial melting and is comparatively simpler. These findings challenge the conventional use of thermoporometry (which assumes a direct link between pore size and calorimetric peak position) for granular powders containing irregular adsorption sites. The study underscores that, in realistic porous media, phase‑transition thermodynamics cannot be reduced to simple geometric relations; instead, a comprehensive model must account for pore‑wall interactions, surface roughness, pressure effects, and memory phenomena arising from previous solid‑liquid cycles.
Comments & Academic Discussion
Loading comments...
Leave a Comment