Quantifying intermittent transport in cell cytoplasm
Active cellular transport is a fundamental mechanism for protein and vesicle delivery, cell cycle and molecular degradation. Viruses can hijack the transport system and use it to reach the nucleus. Most transport processes consist of intermittent dynamics, where the motion of a particle, such as a virus, alternates between pure Brownian and directed movement along microtubules. In this communication, we estimate the mean time for particle to attach to a microtubule network. This computation leads to a coarse grained equation of the intermittent motion in radial and cylindrical geometries. Finally, by using the degradation activity inside the cytoplasm, we obtain refined asymptotic estimations for the probability and the mean time a virus reaches a small nuclear pore.
💡 Research Summary
In this paper the authors address the problem of intracellular transport of particles such as viruses, vesicles, or molecular motors, which typically alternate between periods of free diffusion and directed motion along microtubules (MTs). They formulate the motion as a stochastic process with two distinct regimes: a Brownian regime described by d x = √(2D) dw and a bound regime described by d x = V dt, where D is the diffusion coefficient and V the velocity of motor‑driven transport. The central quantitative goal is to compute the mean first‑passage time (MFPT) for a particle to encounter the MT network, thereby obtaining an analytical expression for the forward binding rate as a function of geometric and kinetic parameters.
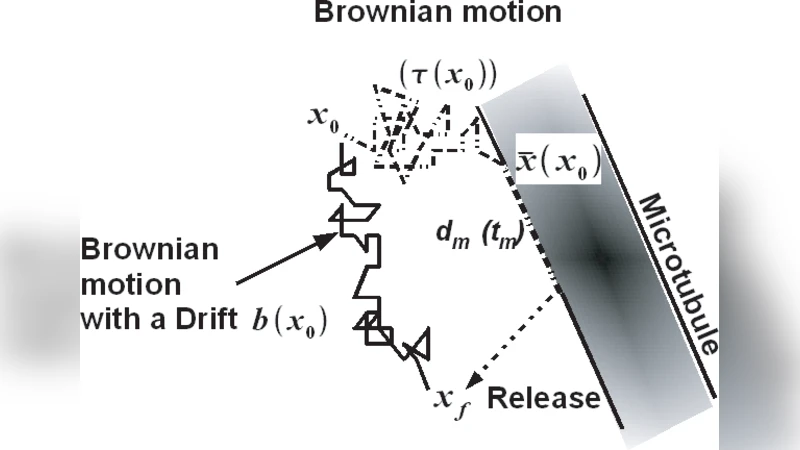
Two idealized geometries are considered. In the two‑dimensional radial model, the cytoplasm is an annulus with outer radius R and inner nuclear radius δ, containing N radially uniformly distributed MTs. The angular spacing between neighboring MTs is Θ = 2π/N, which is assumed to be small. By solving the diffusion problem in a wedge of angle Θ, the authors derive the average time τ(r₀) ≈ r₀²Θ²/(12D) for a particle starting at radius r₀ to hit a MT, and the average binding radius (\bar r(r₀)≈r₀(1+Θ²/12)). After binding, the particle moves a mean distance dₘ = V tₘ (tₘ is the average bound time) toward the nucleus and is released at radius r_f = (\bar r−dₘ). Equating the total intermittent travel time τ(r₀)+tₘ with the mean first‑passage time of an effective drift‑diffusion process yields an explicit effective drift b(r₀) = (dₘ − r₀Θ²/12)/(tₘ + r₀²Θ²/(12D)). This drift replaces the original switching dynamics by a single stochastic differential equation d x = b(x)dt + √(2D)dw.
In the three‑dimensional cylindrical model, a dendrite or axon is approximated as a thin cylinder of radius R and length L, with N parallel MTs modeled as slender cylinders of radius ε ≪ R. The first encounter time with any MT is obtained from the principal eigenvalue λ of the Laplacian with reflecting outer boundary and absorbing MT surfaces: λ ≈ 2πN|Ω| ln(1/ε) where |Ω| = πR². Consequently τ ≈ 1/(λD) ≈ R² ln(1/ε)/(2ND). The effective drift along the axis is then B = dₘ/tₘ + τ, showing a linear dependence on the number of MTs when tₘ ≪ τ.
The authors embed the effective drift into a Fokker‑Planck equation that also includes a spatially dependent killing term k(x) representing enzymatic degradation (e.g., proteases). With reflecting boundaries on the cell membrane and nuclear envelope and an absorbing small target representing a nuclear pore (area fraction ε ≪ 1), they apply small‑hole asymptotics and Laplace’s method to obtain closed‑form approximations for the probability P_N that a particle reaches a pore and the corresponding mean time τ_N. For constant degradation rate k₀ near the nucleus and assuming the drift points inward (b > 0), they find
P_N ≈
Comments & Academic Discussion
Loading comments...
Leave a Comment