Correlated Phenotypic Transitions to Competence in Bacterial Colonies
Genetic competence is a phenotypic state of a bacterial cell in which it is capable of importing DNA, presumably to hasten its exploration of alternate genes in its quest for survival under stress. Recently, it was proposed that this transition is uncorrelated among different cells in the colony. Motivated by several discovered signaling mechanisms which create colony-level responses, we present a model for the influence of quorum-sensing signals on a colony of B. Subtilis cells during the transition to genetic competence. Coupling to the external signal creates an effective inhibitory mechanism, which results in anti-correlation between the cycles of adjacent cells. We show that this scenario is consistent with the specific experimental measurement, which fails to detect some underlying collective signaling mechanisms. Rather, we suggest other parameters that should be used to verify the role of a quorum-sensing signal. We also study the conditions under which phenotypic spatial patterns may emerge.
💡 Research Summary
The paper challenges the prevailing view that the transition to genetic competence in Bacillus subtilis occurs independently in each cell. Building on the well‑established quorum‑sensing (QS) systems of this organism, the authors propose that extracellular signaling molecules act as an effective inhibitor of competence in neighboring cells, thereby generating anti‑correlated competence cycles across the colony.
Model formulation – Each cell i is described by a competence variable (C_i) (high when the cell is in the DNA‑uptake state) and by the local concentration of a QS signal (S_i). The dynamics are captured by two coupled differential equations:
- (\dot C_i = f(C_i) - g,S_i,C_i + \xi_i) – the first term represents the intrinsic positive feedback loop of the competence circuit (ComK/ComS), the second term models inhibition proportional to the external signal, and (\xi_i) is stochastic noise.
- (\dot S_i = \alpha,C_i - \beta,S_i + D\nabla^2 S_i) – competent cells produce signal at rate (\alpha); the signal decays with rate (\beta) and diffuses with coefficient (D).
When the inhibitory coupling constant (g) is positive, a competent cell raises the local signal level, which in turn suppresses competence in its neighbours. This creates a spatial anti‑phase relationship: if one cell is competent, its immediate neighbours are less likely to be competent at the same time.
Simulation outcomes – The authors explore a broad parameter space. Key observations include:
- Wave‑like propagation – Competence events travel across the colony as a travelling wave, with adjacent cells oscillating out of phase.
- Dependence on (g) and (D) – Strong inhibition (large (g)) reduces the overall fraction of competent cells but yields a more uniform spatial distribution. Low diffusion (small (D)) confines the signal, producing localized “spot‑like” clusters where competence is periodically suppressed and re‑emerges.
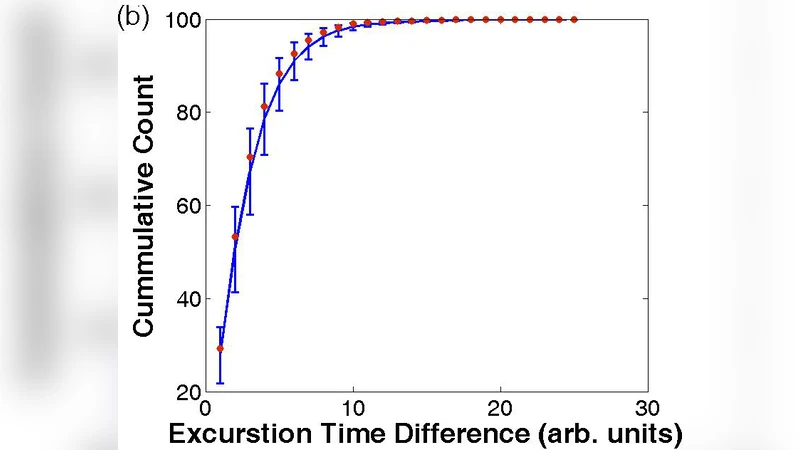
- Agreement with existing data – The mean competence fraction predicted by the model matches experimental measurements that reported a roughly 10 % competent subpopulation. However, the experimentally observed cell‑to‑cell correlation coefficient is near zero. The authors argue that standard fluorescence‑reporter assays only record a binary “competent / non‑competent” state after a fixed delay, thus missing the rapid anti‑correlated oscillations predicted by the model.
Critique of current experimental approaches – The paper points out that GFP‑fusion reporters for ComK become fluorescent only after sustained competence, so short inhibitory pulses are invisible. To capture the predicted dynamics, the authors recommend:
- Real‑time single‑cell tracking of both (C_i) (e.g., using a fast‑maturing fluorescent tag) and extracellular signal concentration (e.g., via a fluorescently labeled peptide or a biosensor strain).
- Microfluidic devices that allow rapid exchange of medium, enabling controlled perturbations of signal levels and direct measurement of the response latency.
- Quantitative assays of signal secretion (mass spectrometry or ELISA) correlated with competence markers on a per‑cell basis.
Pattern formation analysis – By performing linear stability analysis on the coupled equations, the authors identify a Turing‑like instability condition: when the product (g/D) exceeds a critical threshold, spatially periodic patterns of competence can emerge spontaneously. Numerical simulations confirm that, under these conditions, competent cells arrange in quasi‑regular arrays with a characteristic wavelength set by the diffusion length of the signal. This suggests that QS does not merely tune the average competence level but can also impose a higher‑order spatial organization that may help the colony maintain genetic diversity while avoiding excessive DNA uptake that could be deleterious.
Implications and future directions – The study provides a mechanistic framework linking quorum sensing to phenotypic heterogeneity in bacterial colonies. It predicts that anti‑correlated competence cycles are a robust outcome of an inhibitory feedback loop mediated by extracellular signals. To validate the model, future work should:
- Map the full repertoire of competence‑related QS molecules across Bacillus species (e.g., ComX, surfactin, CSF) and determine their respective inhibitory strengths.
- Quantify how environmental stress intensity (nutrient limitation, DNA damage) modulates the production rate (\alpha) and the degradation rate (\beta) of the signal.
- Extend the model to include multiple interacting signals and possible cross‑talk, which could generate richer dynamics such as multistable domains or traveling fronts.
In summary, the paper argues convincingly that the transition to genetic competence is not a purely stochastic, cell‑autonomous event but is shaped by colony‑level quorum‑sensing feedback. By highlighting the limitations of current measurement techniques and proposing concrete experimental strategies, the authors open a path toward uncovering hidden collective behaviors in bacterial populations and exploiting them for synthetic biology, antimicrobial strategies, and evolutionary studies.
Comments & Academic Discussion
Loading comments...
Leave a Comment