Intracellular microrheology of motile Amoeba proteus
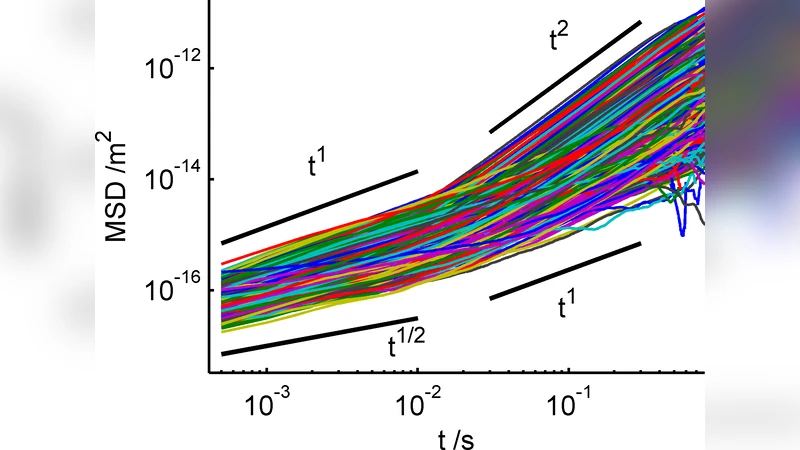
The motility of motile Amoeba proteus was examined using the technique of passive particle tracking microrheology, with the aid of newly-developed particle tracking software, a fast digital camera and an optical microscope. We tracked large numbers of endogeneous particles in the amoebae, which displayed subdiffusive motion at short time scales, corresponding to thermal motion in a viscoelastic medium, and superdiffusive motion at long time scales due to the convection of the cytoplasm. Subdiffusive motion was characterised by a rheological scaling exponent of 3/4 in the cortex, indicative of the semiflexible dynamics of the actin fibres. We observed shear-thinning in the flowing endoplasm, where exponents increased with increasing flow rate; i.e. the endoplasm became more fluid-like. The rheology of the cortex is found to be isotropic, reflecting an isotropic actin gel. A clear difference was seen between cortical and endoplasmic layers in terms of both viscoelasticity and flow velocity, where the profile of the latter is close to a Poiseuille flow for a Newtonian fluid.
💡 Research Summary
The authors employed passive particle‑tracking microrheology to probe the intracellular mechanical environment of the motile protozoan Amoeba proteus. Using a high‑speed digital camera, an optical microscope, and custom‑written tracking software, they recorded the trajectories of thousands of endogenous granules over extended periods. Analysis of the mean‑square displacement (MSD) as a function of lag time revealed two distinct dynamical regimes.
At short lag times (≈10⁻³–10⁻¹ s) the MSD scaled sub‑diffusively with an exponent α≈0.75. This 3/4 scaling is characteristic of thermal fluctuations in a semiflexible polymer network, indicating that the cell cortex behaves as a viscoelastic gel dominated by actin filaments. The exponent’s consistency across many particles and directions demonstrates that the cortical actin mesh is isotropic rather than aligned.
At longer lag times (≥10⁻¹ s) the MSD grew faster than linearly (β > 1), reflecting super‑diffusive transport driven by bulk cytoplasmic flow. By extracting particle velocities, the authors showed that the endoplasmic region exhibits a velocity profile that is parabolic, closely matching Poiseuille flow for a Newtonian fluid. However, the rheological response of this flowing region is not Newtonian: the apparent scaling exponent α increases with flow speed, rising from ~0.78 at low speeds (<5 µm s⁻¹) to ~0.95 at higher speeds (>10 µm s⁻¹). This trend signals shear‑thinning— the actin network becomes more fluid‑like under higher shear rates.
Complex shear moduli G*(ω) were estimated from the MSD data. In the cortex, both storage (G′) and loss (G″) moduli followed ω^0.75 scaling, confirming a semiflexible polymer rheology with a relatively constant loss‑to‑storage ratio (~0.6). In the endoplasm, G′ and G″ decreased with increasing shear rate, producing a classic shear‑thinning curve rather than a constant viscosity.
The study therefore delineates three key mechanical distinctions within a single amoeboid cell: (1) an isotropic, semiflexible actin cortex with a fixed 3/4 scaling exponent, (2) a flowing endoplasm that exhibits Poiseuille‑like velocity distribution but a shear‑dependent viscoelastic response, and (3) a clear transition from sub‑diffusive, thermally driven motion to super‑diffusive, convection‑driven transport as the observation time scale lengthens.
Methodologically, the work showcases how high‑frame‑rate imaging combined with robust particle‑tracking algorithms can resolve intracellular rheology across multiple decades of time, enabling simultaneous measurement of both passive thermal fluctuations and active cytoplasmic streaming. The findings advance our understanding of how amoeboid motility emerges from the interplay between a relatively stiff cortical shell and a more fluid, shear‑thinning interior, and they provide a quantitative framework that can be applied to other motile cells where actin dynamics and cytoplasmic flow are tightly coupled.
Comments & Academic Discussion
Loading comments...
Leave a Comment