Theoretical analyses predict A20 regulates period of NF-kB oscillation
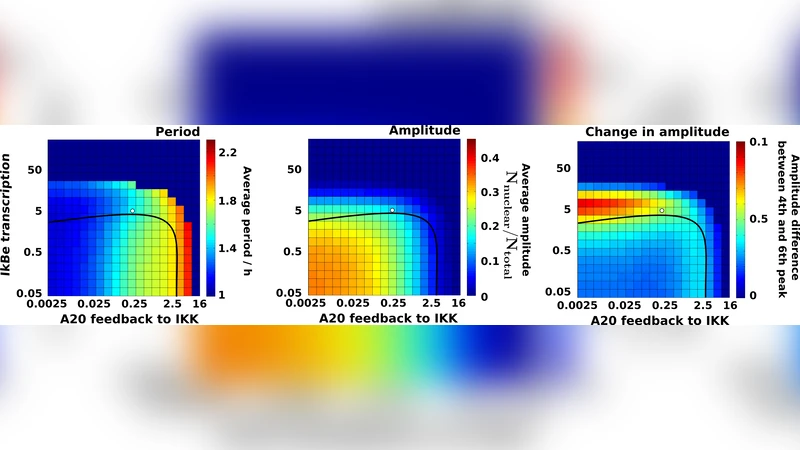
The nuclear-cytoplasmic shuttling of NF-kB is characterized by damped oscillations of the nuclear concentration with a time period of around 1-2 hours. The NF-kB network contains several feedback loops modulating the overall response of NF-kB activity. While IkBa is known to drive and IkBe is known to dampen the oscillations, the precise role of A20 negative feedback remains to be elucidated. Here we propose a model of the NF-kB system focusing on three negative feedback loops (IkBa, IkBe and A20) which capture the experimentally observed responses in wild-type and knockout cells. We find that A20, like IkBe, efficiently dampens the oscillations albeit through a distinct mechanism. In addition, however, we have discovered a new functional role of A20 by which it controls the oscillation period of nuclear NF-kB. The design based on three nested feedback loops allows independent control of period and amplitude decay in the oscillatory response. Based on these results we predict that adjusting the expression level of A20, e.g. by siRNA, the period can be changed by up to a factor 2.
💡 Research Summary
The paper presents a quantitative systems‑biology study of the NF‑κB signaling network, focusing on the damped nuclear‑cytoplasmic oscillations that follow stimulation by pro‑inflammatory cytokines such as TNF‑α. While the rapid negative feedback mediated by IκBα (IkBa) is known to generate oscillations and the slower feedback through IκBε (IkBe) is recognized for damping them, the functional contribution of the ubiquitin‑editing enzyme A20 has remained ambiguous. To resolve this, the authors construct a deterministic ordinary‑differential‑equation (ODE) model that explicitly incorporates three nested negative feedback loops: IkBa, IkBe, and A20. The model contains nine state variables representing nuclear and cytoplasmic NF‑κB, the two IκB‑NF‑κB complexes, A20 mRNA and protein, and active IKK. Each feedback is described by synthesis and degradation terms together with nonlinear saturation (Hill‑type) kinetics, allowing realistic time delays and dose‑response curves.
Parameter values are drawn from the literature and refined by fitting to time‑course data obtained from wild‑type (WT) mouse macrophages and three knockout (KO) lines (IkBa‑KO, IkBe‑KO, A20‑KO) after a 10 ng/mL TNF‑α pulse. Experimental measurements include live‑cell fluorescence imaging of nuclear NF‑κB and Western blot quantification of IκB and A20 levels over an 8‑hour window. The model reproduces the hallmark 1–2 hour period damped oscillation in WT cells, the loss of oscillation in IkBa‑KO (sustained nuclear NF‑κB), the amplified and slower‑decaying oscillation in IkBe‑KO, and a markedly shortened period with reduced amplitude in A20‑KO. These fits validate that the three feedbacks together can capture the full spectrum of observed dynamics.
A key discovery emerges from systematic parameter sweeps of the A20 synthesis rate (k_syn_A20). Reducing k_syn_A20 by 50 % lengthens the oscillation period by roughly a factor of 1.9, whereas doubling k_syn_A20 shortens the period to about 0.8 of the WT value. Importantly, this period modulation occurs with minimal impact on the overall amplitude decay, which remains primarily governed by IkBe. Thus, A20 provides a distinct, slower feedback that can fine‑tune the timing of NF‑κB nuclear excursions without compromising the network’s ability to dampen oscillations. The authors term this “independent control of period and amplitude” and argue that the nested architecture—fast IkBa, intermediate IkBe, and slow A20—creates a modular design space for dynamic regulation.
The discussion extrapolates these findings to physiological contexts. A rapid early oscillation may enhance transcription of immediate‑early genes required for pathogen clearance, while a later A20‑mediated lengthening of the period could help terminate the response and prevent chronic inflammation. The model predicts that experimental manipulation of A20 levels—by siRNA knockdown, CRISPR‑mediated activation, or over‑expression vectors—should shift the NF‑κB period by up to twofold, a hypothesis that can be directly tested in vitro and in vivo. Moreover, the theoretical framework suggests that similar multi‑feedback designs could be employed by other signaling pathways to decouple frequency and amplitude control, offering a general principle for synthetic biology circuit design.
In summary, the study provides a rigorously validated mathematical model that clarifies A20’s dual role: (1) contributing to the damping of NF‑κB oscillations alongside IkBe, and (2) uniquely governing the oscillation period through a slow, transcription‑dependent feedback on IKK activity. These insights not only deepen our mechanistic understanding of NF‑κB dynamics but also open avenues for therapeutic strategies that target A20 to modulate inflammatory timing in diseases such as rheumatoid arthritis, sepsis, and certain cancers.
Comments & Academic Discussion
Loading comments...
Leave a Comment