A model for the generation of localized transient Na+ elevations in vascular smooth muscle
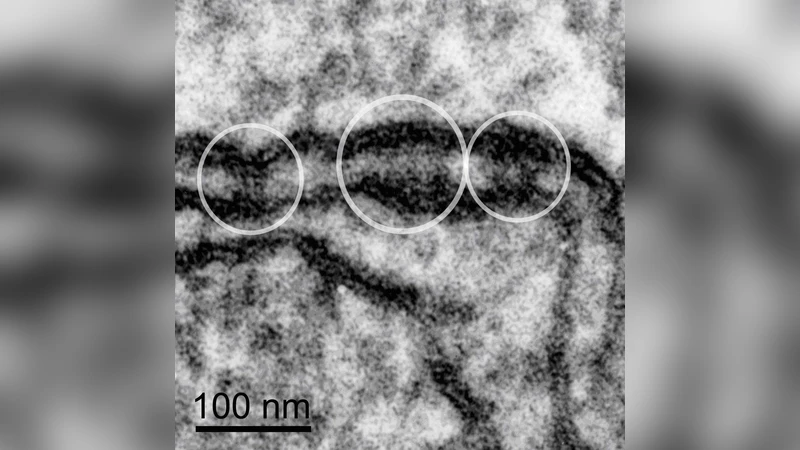
We present a stochastic computational model to study the mechanism of signalling between a source and a target ionic transporter, both localized on the plasma membrane (PM) and in intracellular nanometre-scale subplasmalemmal signalling compartments comprising the PM, the sarcoplasmic reticulum (SR), Ca2+ and Na+ transporters, and the intervening cytosol. We refer to these compartments, sometimes called junctions, as cytoplasmic nanospaces or nanodomains. In the chain of events leading to Ca2+ influx for SR reloading during asynchronous Ca2+ waves in vascular smooth muscle (VSM), the physical and functional link between non-selective cation channels (NSCC) and Na+/Ca2+ exchangers (NCX) needs to be elucidated in view of two recent findings: the identification of the transient receptor potential canonical channel 6 (TRPC6) as a crucial NSCC in VSM cells and the observation of localized cytosolic [Na+] transients following purinergic stimulation of these cells. Having previously helped clarify the Ca2+ signalling step between NCX and SERCA behind SR Ca2+ refilling, this quantitative approach now allows us to model the upstream linkage of NSCC and NCX. We have implemented a random walk (RW) Monte Carlo (MC) model with simulations mimicking a Na+ diffusion process originating at the NSCC within PM-SR junctions. The model calculates the average [Na+] in the nanospace and also produces [Na+] profiles as a function of distance from the Na+ source. Our results highlight the necessity of a strategic juxtaposition of the relevant signalling channels as well as other physical structures within the nanospaces to permit adequate [Na+] build-up to provoke NCX reversal and Ca2+ influx to refill the SR.
💡 Research Summary
The paper addresses a fundamental question in vascular smooth muscle (VSM) physiology: how non‑selective cation channels (NSCC), specifically TRPC6, communicate with the Na⁺/Ca²⁺ exchanger (NCX) to trigger Ca²⁺ influx that refills the sarcoplasmic reticulum (SR) during asynchronous Ca²⁺ waves. Recent experimental observations have identified TRPC6 as the dominant NSCC in VSM cells and have reported localized cytosolic Na⁺ transients following purinergic stimulation. While previous work clarified the downstream Ca²⁺ transfer from NCX to SERCA, the upstream coupling between NSCC and NCX remained speculative. To fill this gap, the authors constructed a stochastic, three‑dimensional Monte Carlo random‑walk model that simulates Na⁺ diffusion from a point source (the open TRPC6 channel) within a nanometer‑scale junction formed by the plasma membrane (PM), the sub‑plasmalemmal sarcoplasmic reticulum (SR), and the intervening cytosol.
The geometric core of the model is a nanodomain of approximately 100 nm × 100 nm in lateral dimensions and a 20 nm gap between PM and SR, reflecting electron‑microscopy measurements of VSM junctions. This volume is discretized into a cubic lattice with a 0.2 nm spacing, providing a high‑resolution grid for tracking individual Na⁺ ions. The diffusion coefficient used (D ≈ 1.33 × 10⁻⁹ m² s⁻¹) matches experimental values for Na⁺ in cytosol. Boundary conditions are set such that the PM and SR surfaces act as reflective walls (preventing ions from leaving the junction), while the outer boundary of the nanodomain is absorptive, mimicking ion loss into the bulk cytoplasm.
In each simulation step, a Na⁺ ion moves randomly to one of its six neighboring lattice points, reproducing a classic random‑walk process. The source term is defined by a Poisson process that injects ions at a rate consistent with measured TRPC6 conductance (≈10⁶ ions s⁻¹). By running the walk for up to 1 ms (10⁶ steps), the model generates spatial Na⁺ concentration profiles and temporal averages. The authors systematically varied key parameters: source‑to‑NCX distance (10–50 nm), presence of immobile obstacles (representing cytoskeletal proteins or other membrane proteins), and channel open‑time patterns (continuous versus brief pulses).
The simulations reveal several crucial points. First, when the NSCC and NCX are positioned within 10–30 nm, the confined geometry and reflective boundaries cause Na⁺ to accumulate rapidly, reaching local concentrations of 15–20 mM. This level exceeds the threshold (~10 mM) required for NCX reversal, where the exchanger switches from exporting Ca²⁺ to importing it, thereby providing the Ca²⁺ influx needed for SR refilling. Second, the inclusion of static obstacles within the nanodomain further restricts diffusion pathways, sharpening the Na⁺ concentration peak and extending its lifetime. This finding aligns with the notion that protein scaffolds or actin filaments may act as diffusion barriers in vivo. Third, the model demonstrates that sustained channel opening (>10 ms) yields the highest Na⁺ peaks, yet physiologically realistic brief openings (1–5 ms) repeated at 20 ms intervals also achieve sufficient accumulation for NCX reversal. Thus, transient purinergic signals can be effective without requiring prolonged depolarization.
Parameter sweeps also predict how pathological alterations could affect signaling. Enlarged nanogaps (30–50 nm) as observed in hypertension or diabetes reduce Na⁺ buildup, potentially impairing NCX‑mediated Ca²⁺ entry and compromising SR refilling. Conversely, up‑regulation of TRPC6 expression or increased open probability amplifies Na⁺ accumulation, which may contribute to hypercontractile states. The model therefore provides a quantitative framework for testing therapeutic interventions that target either the spatial arrangement of channels (e.g., nanodomain‑stabilizing agents) or the functional properties of TRPC6 (e.g., selective blockers).
In summary, the authors present a rigorously validated stochastic diffusion model that links TRPC6‑derived Na⁺ influx to NCX reversal within sub‑plasmalemmal nanodomains. The work emphasizes that not only electrical proximity but also precise spatial juxtaposition and structural confinement are essential for generating the localized Na⁺ transients observed experimentally. By bridging molecular biophysics with cellular physiology, the study offers a powerful tool for exploring VSM signaling under both normal and disease conditions, and it suggests new avenues for pharmacological modulation of vascular tone.
Comments & Academic Discussion
Loading comments...
Leave a Comment