Sequence reproduction, single trial learning, and mimicry based on a mammalian-like distributed code for time
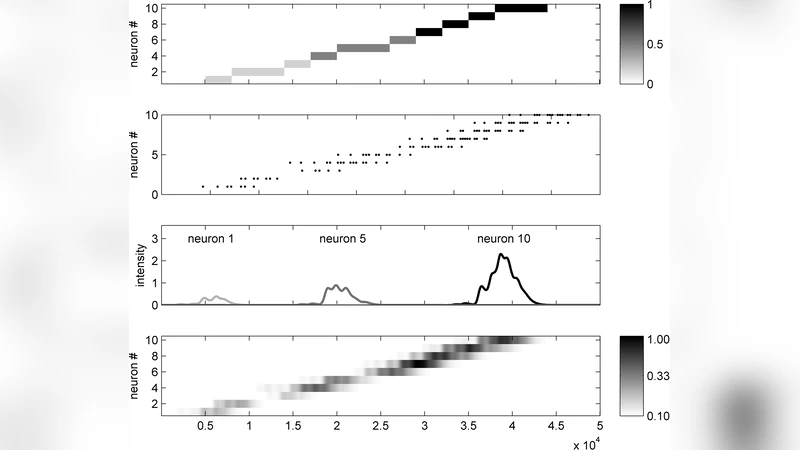
Animals learn tasks requiring a sequence of actions over time. Waiting a given time before taking an action is a simple example. Mimicry is a complex example, e.g. in humans, humming a brief tune you have just heard. Re-experiencing a sensory pattern mentally must involve reproducing a sequence of neural activities over time. In mammals, neurons in prefrontal cortex have time-dependent firing rates that vary smoothly and slowly in a stereotyped fashion. We show through modeling that a Many are Equal computation can use such slowly-varying activities to identify each timepoint in a sequence by the population pattern of activity at the timepoint. The MAE operation implemented here is facilitated by a common inhibitory conductivity due to a theta rhythm. Sequences of analog values of discrete events, exemplified by a brief tune having notes of different durations and intensities, can be learned in a single trial through STDP. An action sequence can be played back sped up, slowed down, or reversed by modulating the system that generates the slowly changing stereotyped activities. Synaptic adaptation and cellular post-hyperpolarization rebound contribute to robustness. An ability to mimic a sequence only seconds after observing it requires the STDP to be effective within seconds.
💡 Research Summary
**
The paper addresses how mammals can learn and reproduce temporally ordered actions, ranging from simple waiting periods to complex mimicry such as humming a short tune after hearing it. The authors build on the observation that neurons in the prefrontal cortex (PFC) exhibit slowly varying, smooth firing‑rate trajectories that are stereotyped across trials. They propose that these continuous “time‑dependent” firing patterns can serve as a distributed temporal code, allowing the brain to identify each moment in a sequence by the population activity pattern present at that moment.
Central to the model is a “Many‑Are‑Equal” (MAE) computation. In the MAE operation, a set of neurons whose instantaneous firing rates are sufficiently similar trigger a common inhibitory conductance. This conductance is generated by a shared inhibitory synapse that receives a rhythmic drive at theta frequency (4–8 Hz). The theta rhythm synchronizes the inhibitory bursts, effectively sampling the population code at regular intervals and producing a sharp “time‑stamp” signal whenever the population pattern matches a stored template.
Learning proceeds in a single trial via spike‑timing‑dependent plasticity (STDP). When an external sensory event (e.g., a note of a particular pitch, intensity, and duration) arrives a few milliseconds before the MAE‑generated inhibitory pulse, the synapse linking the sensory input to the downstream read‑out neuron is potentiated; if it arrives after the inhibitory pulse, the synapse is depressed. Because the temporal offset required for potentiation is on the order of 10–30 ms, the STDP window is narrow enough to allow a single exposure to create a robust association between the sensory pattern and the corresponding time‑stamp. Consequently, the system can store an entire sequence of analog values (the notes of a tune) after a single listening episode.
During replay, the slowly varying background activity is left intact while external sensory drive is removed. The stored synaptic weights cause the MAE‑generated inhibitory pulses to reactivate the appropriate downstream neurons at the correct moments, recreating the original sequence. By modulating the speed of the underlying background dynamics, the replay can be sped up, slowed down, or even run in reverse. The model also incorporates synaptic adaptation (short‑term depression) and post‑hyperpolarization rebound of neurons, which together increase robustness against noise, fatigue, and variability in the background rhythm.
Simulation results with a network of 100 model neurons demonstrate that a five‑note melody can be learned in a single exposure, then reproduced accurately without external input. Changing the background speed by factors of 0.5 and 2 preserves the relative timing and intensity of the notes, while reversing the direction of the background dynamics yields a perfect backward replay. Adding stochastic perturbations to firing rates reduces performance only modestly, confirming the system’s tolerance to biological variability.
The authors compare their approach to traditional clock‑based or token‑based models of sequence learning. Unlike those models, which rely on discrete timers or external clock signals, the present framework exploits an intrinsic, continuously evolving neural trajectory that is already present in the cortex. This makes the mechanism biologically plausible and aligns it with experimentally observed PFC dynamics.
In summary, the paper proposes that a mammalian‑like distributed temporal code, combined with a theta‑synchronized inhibitory “Many‑Are‑Equal” operation and rapid STDP, can account for single‑trial acquisition, flexible speed modulation, reversal, and noise‑robust replay of complex action sequences such as humming a newly heard tune. The findings offer a concrete computational hypothesis for how the brain internally reproduces sensory patterns and suggest new design principles for artificial neural systems that need to learn and reproduce temporal sequences from minimal exposure.
Comments & Academic Discussion
Loading comments...
Leave a Comment