Arginine-rich peptides destabilize the plasma membrane, consistent with a pore formation translocation mechanism of cell penetrating peptides
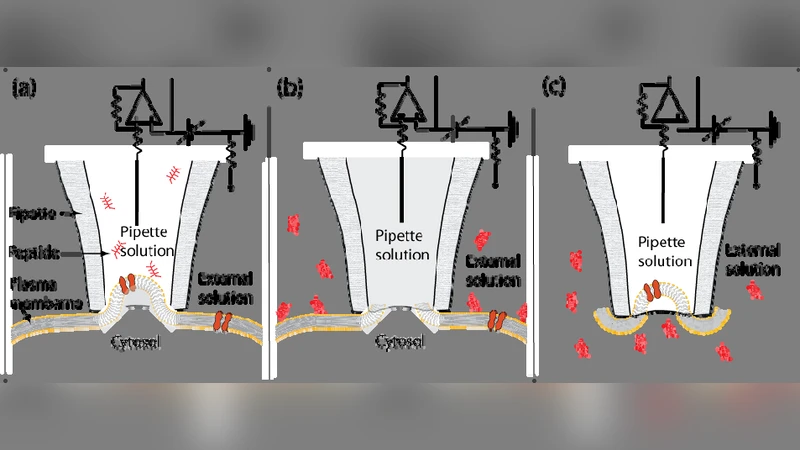
Recent molecular dynamics simulations (Herce and Garcia, PNAS, 104: 20805 (2007)) have suggested that the arginine-rich HIV Tat peptides might be able to translocate by destabilizing and inducing transient pores in phospholipid bilayers. In this pathway for peptide translocation, arginine residues play a fundamental role not only in the binding of the peptide to the surface of the membrane but also in the destabilization and nucleation of transient pores across the bilayer, despite being charged and highly hydrophilic. Here we present a molecular dynamics simulation of a peptide composed of nine arginines (Arg-9) that shows that this peptide follows the same translocation pathway previously found for the Tat peptide. We test this hypothesis experimentally by measuring ionic currents across phospholipid bilayers and cell membranes through the pores induced by Arg-9 peptides. We find that Arg-9 peptides, in the presence of an electrostatic potential gradient, induce ionic currents across planar phospholipid bilayers, as well as in cultured osteosarcoma cells and human smooth muscle cells freshly isolated from the umbilical artery. Our results suggest that the mechanism of action of Arg-9 peptide involves the creation of transient pores in lipid bilayers and cell membranes.
💡 Research Summary
The paper investigates how arginine‑rich peptides, specifically a nona‑arginine (Arg‑9) sequence, can traverse lipid membranes by destabilizing them and forming transient pores. Building on earlier molecular dynamics (MD) simulations that suggested the HIV Tat peptide uses a pore‑formation mechanism, the authors extend the hypothesis to a peptide composed solely of nine consecutive arginine residues.
In the computational part, the authors performed all‑atom MD simulations using the CHARMM36 force field on a POPC bilayer under an applied transmembrane voltage of ±150 mV. Arg‑9 was initially placed near the membrane surface. The simulations revealed rapid electrostatic attraction of the positively charged guanidinium groups to the negatively charged phosphate headgroups, leading to a network of hydrogen bonds and a local thinning of the bilayer. The side chains of arginine, being long and highly charged, recruit water molecules and create a hydrated conduit. Over ~200 ns, this conduit expands into a continuous water‑filled channel with an estimated diameter of 0.5–1 nm and a length comparable to the bilayer thickness. When the voltage is removed, the channel collapses, indicating that the pore is voltage‑dependent and transient.
To validate the computational predictions, the authors measured ionic currents across planar lipid bilayers and across the plasma membranes of two cell types: cultured human osteosarcoma (U2‑OS) cells and freshly isolated human umbilical artery smooth muscle cells. In the planar bilayer experiments, addition of 10 µM Arg‑9 under a 100–150 mV potential produced stepwise increases in current of 10–30 pA. The current direction reversed with voltage polarity and vanished when the voltage was turned off, confirming that the peptide induces a voltage‑gated conductive pathway.
In cellular recordings, whole‑cell patch clamp showed that Arg‑9, applied in the presence of a ±150 mV extracellular field, generated transient currents of 20–50 pA in the voltage‑clamped cells, whereas no current was observed in control cells lacking the field. Membrane resistance decreased during peptide exposure and recovered after the field was removed, consistent with the formation of reversible pores.
The authors discuss the mechanistic implications: the high charge density of a poly‑arginine chain creates a strong electrostatic interaction with the membrane surface, storing energy that can be released as a mechanical perturbation when an external electric field is present. This perturbation nucleates a pore, allowing ions—and potentially the peptide itself—to cross the barrier without requiring large conformational changes in the peptide (as posited by direct‑penetration models). The data also suggest a cooperative effect: a single arginine residue does not destabilize the membrane appreciably, but a stretch of nine residues provides sufficient charge to overcome the energetic barrier for pore nucleation.
Overall, the study provides a coherent picture that combines atomistic simulations with electrophysiological measurements to demonstrate that Arg‑9 can destabilize phospholipid bilayers and create transient, voltage‑dependent pores. This mechanism likely underlies the cell‑penetrating activity of many arginine‑rich peptides and offers a rational basis for designing more efficient delivery vectors for therapeutic cargos, gene‑editing tools, or imaging agents.
Comments & Academic Discussion
Loading comments...
Leave a Comment