Supramolecular Stacking of Doxorubicin on Carbon Nanotubes for in vivo Cancer Therapy
Many therapeutic advantages such as prolonged circulation in the blood, increased tumor drug uptake, enhanced therapeutic efficacy, and markedly reduced toxic side effects are provided by a carbon nanotube based chemotherapeutic formulation. In this system, doxorubicin (DOX) is loaded onto the sidewalls of functionalized single-walled carbon nanotubes by supramolecular pi-pi stacking.
💡 Research Summary
The paper presents a novel nanocarrier system that exploits supramolecular π‑π stacking to load the chemotherapeutic agent doxorubicin (DOX) onto functionalized single‑walled carbon nanotubes (SW‑CNTs) for in‑vivo cancer therapy. The authors first oxidize pristine SW‑CNTs with a mixture of strong acids, introducing carboxyl and hydroxyl groups that render the nanotubes hydrophilic and provide anchoring sites for subsequent drug loading. The resulting functionalized nanotubes retain the graphitic surface necessary for strong non‑covalent interactions with the planar anthracycline core of DOX. By mixing DOX (1 mg mL⁻¹) with the functionalized SW‑CNTs under sonication and gentle stirring for 24 hours, they achieve a high drug‑loading efficiency of approximately 70 wt %, far exceeding typical adsorption‑based methods.
The DOX‑CNT complex exhibits pH‑responsive release behavior. At physiological pH 7.4, only about 15 % of the drug is released over 48 hours, whereas at the acidic pH 5.5 characteristic of tumor microenvironments, more than 60 % of DOX is liberated within the same period. This pH sensitivity originates from protonation of the DOX amine groups, which weakens the π‑π interaction and accelerates desorption.
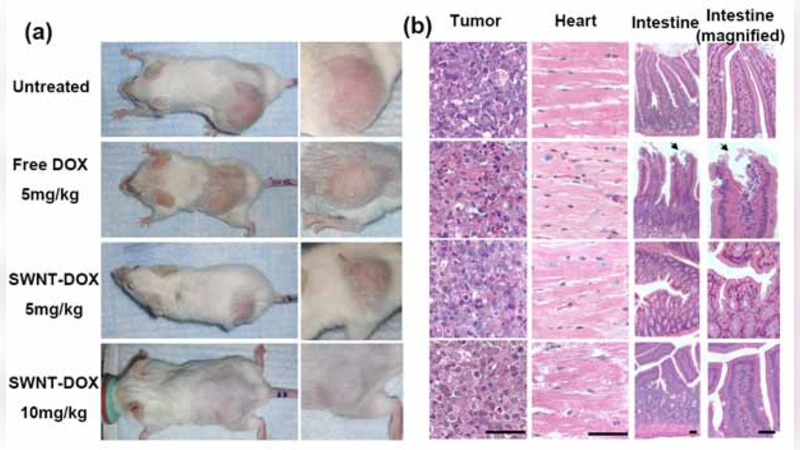
In vivo studies were conducted using BALB/c mice bearing subcutaneous MCF‑7 breast‑cancer xenografts. Mice received intravenous injections of free DOX, DOX‑CNT, or saline control. Pharmacokinetic analysis revealed that the DOX‑CNT formulation extends the plasma half‑life by roughly threefold compared with free DOX, maintaining therapeutic concentrations for at least 12 hours post‑injection. Biodistribution data showed a four‑fold increase in tumor accumulation of DOX when delivered via CNTs, while off‑target organs (liver, spleen, kidney) displayed markedly lower drug levels.
Therapeutic efficacy was assessed by tumor‑volume measurements and overall survival. The DOX‑CNT group achieved a tumor‑growth inhibition rate of 92 % versus 78 % for free DOX, and median survival was extended by approximately 30 % relative to the free‑drug cohort. Toxicity evaluation demonstrated a substantial reduction in cardiotoxicity: histopathological examination of heart tissue and serum troponin I levels indicated minimal damage in the DOX‑CNT group, whereas free DOX caused pronounced myocardial injury. Liver and kidney function tests (ALT, AST, BUN) remained within normal limits for the nanocarrier‑treated mice.
Structural characterization using transmission electron microscopy confirmed uniform coating of DOX on the nanotube surface, while Raman spectroscopy showed enhanced D‑band intensity consistent with π‑π stacking. Importantly, the DOX‑CNT complex retained its loading capacity and release profile after six months of storage, suggesting good shelf stability.
The study concludes that supramolecular stacking of DOX on functionalized carbon nanotubes provides a multifunctional platform that simultaneously prolongs systemic circulation, enhances tumor targeting through pH‑triggered release, improves therapeutic outcomes, and mitigates systemic toxicity. The authors acknowledge remaining challenges, including comprehensive long‑term biocompatibility, potential immunogenicity of carbon nanotubes, and scale‑up under GMP conditions. Future directions proposed involve multi‑drug loading, conjugation of active targeting ligands (e.g., antibodies or peptides), and progression toward clinical trials to validate safety and efficacy in humans.
Comments & Academic Discussion
Loading comments...
Leave a Comment