A Model of Sequential Branching in Hierarchical Cell Fate Determination
Multipotent stem or progenitor cells undergo a sequential series of binary fate decisions, which ultimately generate the diversity of differentiated cells. Efforts to understand cell fate control have focused on simple gene regulatory circuits that predict the presence of multiple stable states, bifurcations and switch-like transitions. However, existing gene network models do not explain more complex properties of cell fate dynamics such as the hierarchical branching of developmental paths. Here, we construct a generic minimal model of the genetic regulatory network controlling cell fate determination, which exhibits five elementary characteristics of cell differentiation: stability, directionality, branching, exclusivity, and promiscuous expression. We argue that a modular architecture comprising repeated network elements reproduces these features of differentiation by sequentially repressing selected modules and hence restricting the dynamics to lower dimensional subspaces of the high-dimensional state space. We implement our model both with ordinary differential equations (ODEs), to explore the role of bifurcations in producing the one-way character of differentiation, and with stochastic differential equations (SDEs), to demonstrate the effect of noise on the system. We further argue that binary cell fate decisions are prevalent in cell differentiation due to general features of the underlying dynamical system. This minimal model makes testable predictions about the structural basis for directional, discrete and diversifying cell phenotype development and thus can guide the evaluation of real gene regulatory networks that govern differentiation.
💡 Research Summary
The paper addresses a fundamental question in developmental biology: how multipotent stem or progenitor cells generate a diverse array of differentiated cell types through a series of binary fate choices. While many previous studies have modeled cell‑fate decisions using simple bistable gene‑regulatory circuits, those models fall short of capturing the hierarchical branching that characterizes real developmental lineages. To fill this gap, the authors construct a generic, minimal model built from repeatedly arranged network modules. Each module consists of two mutually repressing transcription factors that also self‑activate, forming a classic toggle‑switch motif. Crucially, modules are linked in a hierarchical fashion: the activity of an upstream module represses the downstream module’s expression, thereby restricting the system’s dynamics to progressively lower‑dimensional subspaces as differentiation proceeds.
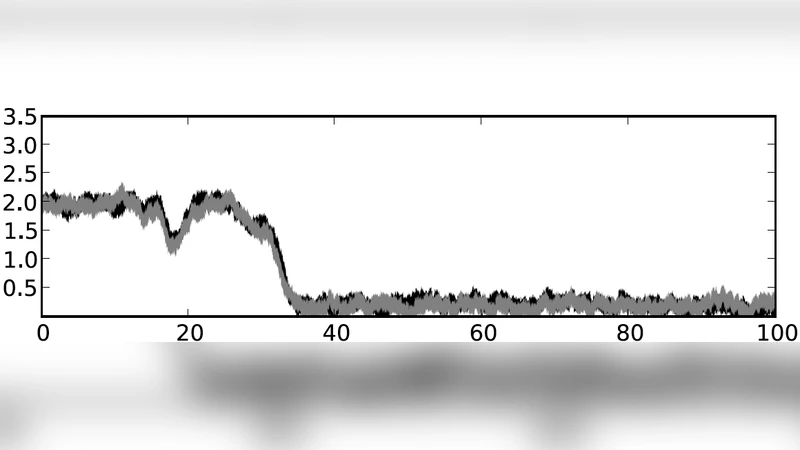
Mathematically the model is expressed as a set of ordinary differential equations (ODEs) for each module’s two variables (x_i, y_i). The ODEs contain nonlinear terms for self‑activation, mutual inhibition, and repression from the preceding module. By varying a single control parameter that represents the strength of upstream repression, the system undergoes a sequence of subcritical pitchfork bifurcations. At each bifurcation the two stable fixed points of the current module disappear, leaving only the fixed points of the next module. This creates a one‑way transition: once a module is silenced, the cell cannot revert to the previous state, establishing directionality. The bifurcation also generates a branch point, because the downstream module presents two alternative stable states, thereby reproducing the binary branching observed in lineage trees.
The authors identify five elementary properties of differentiation that emerge naturally from this architecture: (1) stability – each differentiated fate corresponds to a stable attractor; (2) directionality – the cascade of bifurcations forces irreversible progression; (3) branching – each step offers a binary choice, generating a hierarchical tree; (4) exclusivity – mutually exclusive expression of fate‑specific genes is enforced by cross‑module repression; and (5) promiscuous expression – transient co‑expression of genes from adjacent modules occurs during the transition. To explore the impact of molecular noise, the deterministic ODE framework is extended to stochastic differential equations (SDEs) by adding Gaussian white‑noise terms. Simulations reveal that noise can accelerate transitions, broaden the window of co‑expression, and occasionally cause rare “back‑sliding” events, but the overall hierarchical structure remains robust.
A key conceptual contribution is the explanation of why binary decisions dominate developmental processes. The hierarchical modular design reduces the effective dimensionality by one at each step, leaving only two viable states for the next decision. This dimensional reduction makes binary switches both mathematically stable and evolutionarily economical compared with multi‑branch switches that would require finely tuned parameter spaces.
The paper also outlines testable predictions. Perturbing a specific module—either by over‑expressing one transcription factor or by knocking down the repressive link—should disrupt the expected dimensional reduction, leading to abnormal co‑expression patterns or failure to commit to downstream fates. Single‑cell RNA‑seq data could be mined to verify the predicted sequential silencing of upstream modules and the emergence of lower‑dimensional expression manifolds at each branch point.
In summary, the authors provide a concise yet powerful dynamical systems model that captures the essential features of hierarchical cell‑fate determination. By demonstrating how repeated toggle‑switch modules, coupled through hierarchical repression, generate stability, directionality, branching, exclusivity, and transient promiscuity, the work offers a unifying framework that can guide the analysis of real gene‑regulatory networks and inspire synthetic biology approaches to engineer controlled differentiation pathways.
Comments & Academic Discussion
Loading comments...
Leave a Comment