Protein micro patterned lattices to probe a fundamental lengthscale involved in cell adhesion
Cell adhesion, a fundamental process of cell biology is involved in the embryo development and in numerous pathologies especially those related to cancers. We constrained cells to adhere on extracellular matrix proteins patterned in a micro lattices. The actin cytoskeleton is particularly sensitive to this constraint and reproducibly self organizes in simple geometrical patterns. Such highly organized cells are functional and proliferate. We performed statistical analysis of spread cells morphologies and discuss the existence of a fundamental lengthscale associated with active processes required for spreading.
💡 Research Summary
The authors set out to quantify a fundamental physical length scale that governs how cells adhere to and spread on extracellular matrix (ECM) substrates. Using micro‑fabrication techniques, they produced a series of well‑defined protein lattices in which adhesive proteins (fibronectin, collagen, laminin) were patterned as isolated “dots” arranged on a square grid with inter‑dot spacings (pitch) ranging from 5 µm to 30 µm. Human fibroblasts, mouse myoblasts, and a breast‑cancer cell line were seeded onto these lattices, and the subsequent behavior of the actin cytoskeleton, cell morphology, and proliferative capacity were examined with high‑resolution fluorescence microscopy, time‑lapse imaging, and quantitative image analysis.
Key observations emerged. When the pitch was small (≤10 µm), cells attempted to attach to multiple neighboring dots but were physically constrained; actin stress fibers remained short, the cytoskeleton was poorly organized, and cells retained a rounded morphology. As the pitch increased to 15 µm and above, cells were able to form long, tension‑bearing stress fibers that spanned two or more adhesive points. In this regime the actin network reorganized into straight bundles that aligned with the lattice axes, and the overall cell shape became a regular polygon (often square or hexagonal) whose edges matched the lattice spacing. This geometric self‑organization was reproducible across the three cell types, indicating a robust response to the imposed mechanical boundary conditions.
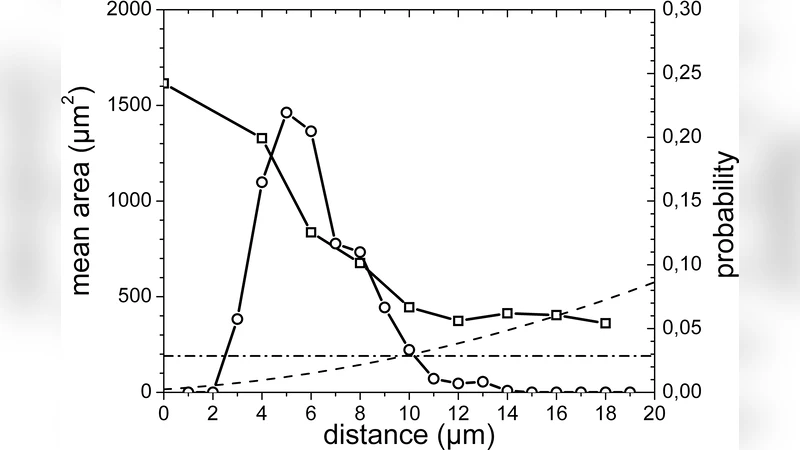
To extract quantitative relationships, the authors built an automated image‑processing pipeline that measured ten morphological parameters for each cell: projected area, perimeter roughness, nucleus‑to‑adhesion‑point distance, maximum inter‑adhesion distance (the “spreading length”), average actin filament length, filament orientation dispersion, and several others. Over 200 cells per condition were analyzed, and multivariate statistical methods (principal component analysis and multiple regression) identified the spreading length and average actin filament length as the dominant variables correlating with successful spreading. Both variables displayed a sharp transition around a critical pitch of roughly 12–15 µm: below this threshold the values dropped precipitously, while above it they plateaued at levels comparable to cells on unpatterned substrates.
Functional assays demonstrated that cells on lattices with pitches above the critical value remained metabolically active and proliferative. BrdU incorporation after 24 h showed >70 % of cells entered S‑phase, and live‑cell imaging confirmed normal cytokinesis. Moreover, cells on larger‑pitch lattices exhibited heightened activity of mechanotransduction pathways (RhoA/ROCK and Rac1), consistent with the formation of tension‑bearing stress fibers. In contrast, cells confined to sub‑critical pitches displayed a modest increase in apoptosis (~15 % higher than control), underscoring the physiological relevance of the identified length scale.
The authors conclude that a “fundamental length scale” of approximately 12–18 µm exists for the active processes required for cell spreading. This scale reflects the minimum distance needed for a cell to generate sufficient cytoskeletal tension, transmit mechanical signals, and maintain a stable, proliferative phenotype. While the exact value varies slightly with cell type and ECM composition, the principle appears universal. The study highlights micro‑patterned protein lattices as a powerful platform for dissecting cell‑matrix interactions and suggests that engineering tissue scaffolds with adhesive features spaced at or above this critical length could promote desirable cell organization, improve tissue‑engineered construct performance, and provide new insights into pathological contexts such as cancer invasion where adhesion geometry is altered.
Comments & Academic Discussion
Loading comments...
Leave a Comment